An analysis is presented of delivery room (DR) neonatal resuscitation practices in Spanish hospitals.
MethodsA questionnaire was sent by e-mail to all hospitals attending deliveries in Spain.
ResultsA total of 180 questionnaires were sent, of which 155 were fully completed (86%). Less than half (71, 46%) were level I or II hospitals, while 84 were level III hospital (54%). In almost three-quarters (74.2%) of the centres, parents and medical staff were involved in the decision on whether to start resuscitation or withdraw it. A qualified resuscitation team (at least two members) was available in 80% of the participant centres (63.9% level I–II, and 94.0% level III, P<.001). Neonatal resuscitation courses were held in 90.3% of the centres. The availability of gas blenders, pulse oximeters, manual ventilators, and plastic wraps was higher in level III hospitals. Plastic wraps for pre-term hypothermia prevention were used in 63.9% of the centres (40.8% level I–II and 83.3% level III, P<.001). Term newborn resuscitation was started on room air in 89.7% of the centres. A manual ventilator (T-piece) was the device used in most cases when ventilation was required (42.3% level I–II and 78.6% level III, P<.001). Early CPAP in preterm infants was applied in 91.7% of the tertiary hospitals. In the last 5 years some practices have improved, such as neonatal resuscitation training, pulse oximeter use, or early CPAP support.
ConclusionsThere is an improvement in some practices of neonatal resuscitation. Significant differences have been found regarding the equipment or practices in the DR, when comparing hospitals of different levels of care.
Se analizan prácticas de reanimación neonatal en salas de partos (SP) de centros hospitalarios españoles.
MétodosSe envió un cuestionario por centro a neonatólogos responsables de la atención del RN en SP de hospitales españoles.
ResultadosDe 180 cuestionarios enviados, se cumplimentaron 155 (86%); 71 centros fueron de nivel I-II (46%) y 84 de nivel III (54%). La familia y el equipo médico participaron en decisiones de no reanimar o interrumpir la reanimación en el 74,2% de los centros. La disponibilidad de 2 o más reanimadores fue del 80% (94,0% en nivel II I y 63,9% en nivel I-II, p < 0,001). En un 90,3% de centros se realizan cursos de Reanimación. En centros de nivel III fueron más frecuentes los mezcladores de gases, pulsioxímetros, ventiladores manuales y envoltorios de plástico. El uso de envoltorios de polietileno fue del 63,9%. En RN a término se inició la reanimación con aire en el 89,7% de los centros. El dispositivo más usado para aplicar VPP fue el «ventilador manual» (78,6% en nivel III y 42,3% en nivel I-II, p < 0,001). En el 91,7% de los centros de nivel III se utilizó CPAP precoz en prematuros. En los últimos 5 años han mejorado prácticas como son la formación de profesionales, el uso de pulsioxímetros y de CPAP precoz.
ConclusionesExiste una mejora progresiva en algunas prácticas de reanimación neonatal. Se encuentran diferencias en aspectos generales, equipamientos y protocolos de actuación durante la reanimación y transporte entre unidades de diferentes niveles.
Stabilisation and resuscitation at birth continue to be universal medical practices and it is estimated that one quarter of neonatal deaths are caused by birth asphyxia.1
Different organisations, such as the International Liaison Committee on Resuscitation (ILCOR),2 the European Resuscitation Council,3 the American Heart Association4 and the Australian and New Zealand Resuscitation Councils5 have published recommendations for neonatal resuscitation on a regular basis. Since its inception, the Group on Neonatal Resuscitation of the SENeo (Grupo de Reanimación Neonatal de la SENeo [GRN-SENeo]) has disseminated these recommendations through a manual that is now on its third edition6 in addition to informative articles.7
While there is consensus between institutions, neonatal resuscitation practices vary across countries and even between hospitals within a single country.8–13
In 2009, the GRN-SENeo published a survey on neonatal resuscitation in delivery rooms that summarised the practices of the major Spanish hospitals in 2007.14 Following the publication of the 2010 recommendations2–4 and the third edition of the Spanish Manual de reanimación neonatal (Manual of neonatal resuscitation),7 the GRN-SENeo designed a new survey with the objective of assessing the evolution of resuscitation practices in Spain. This article presents and discusses the main results obtained from this assessment and compares them with the practices of other countries.
Materials and methodsIn the last trimester of 2012, we sent a questionnaire by electronic mail to the major hospitals that attend deliveries in Spain. The questionnaire consisted of 48 items that explored aspects related to resuscitation in the delivery room: hospital demographics, ethics, equipment and materials, staff availability and qualifications, resuscitation clinical practices and care after resuscitation (see Appendix B).
We performed a descriptive statistical analysis, expressing qualitative variables as absolute and relative frequencies. We have compared qualitative variables by means of the χ2 test. We considered P-values of 0.05 or less to be statistically significant. We show overall results for different aspects of resuscitation, comparing participating hospitals by level of care (Table 1).
Neonatal unit levels of care.
| Level I | Level II-A | Level II-B | Level III-A | Level III-B | Level III-C |
|---|---|---|---|---|---|
| NBs with GA>35 weeks Multiple gestations (Maximum 2 foetuses) N deliveries: <1000/year | NBs with GA>32 weeks and weight>1500g Back transfer from NICU | Oxygen therapy and CPAP 24-h mechanical ventilation Partial EXT Parenteral nutrition | NBs with GA>28 weeks and weight>1000g Prolonged mechanical ventilation Total EXT Peritoneal dialysis Thoracocentesis Minor surgery | NBs with GA<28 weeks and weight<1000g HFOV and nitric oxide Advanced diagnostic imaging Major surgery | Haemofiltration and haemodialysis Heart surgery with ECC ECMO |
ECC, extracorporeal circulation; CPAP, continuous positive airway pressure; GA, gestational age; ECMO, extracorporeal membrane oxygenation; EXT, exchange transfusion; NB, newborn; NICU, neonatal intensive care unit; HFOV, high frequency oscillatory ventilation.
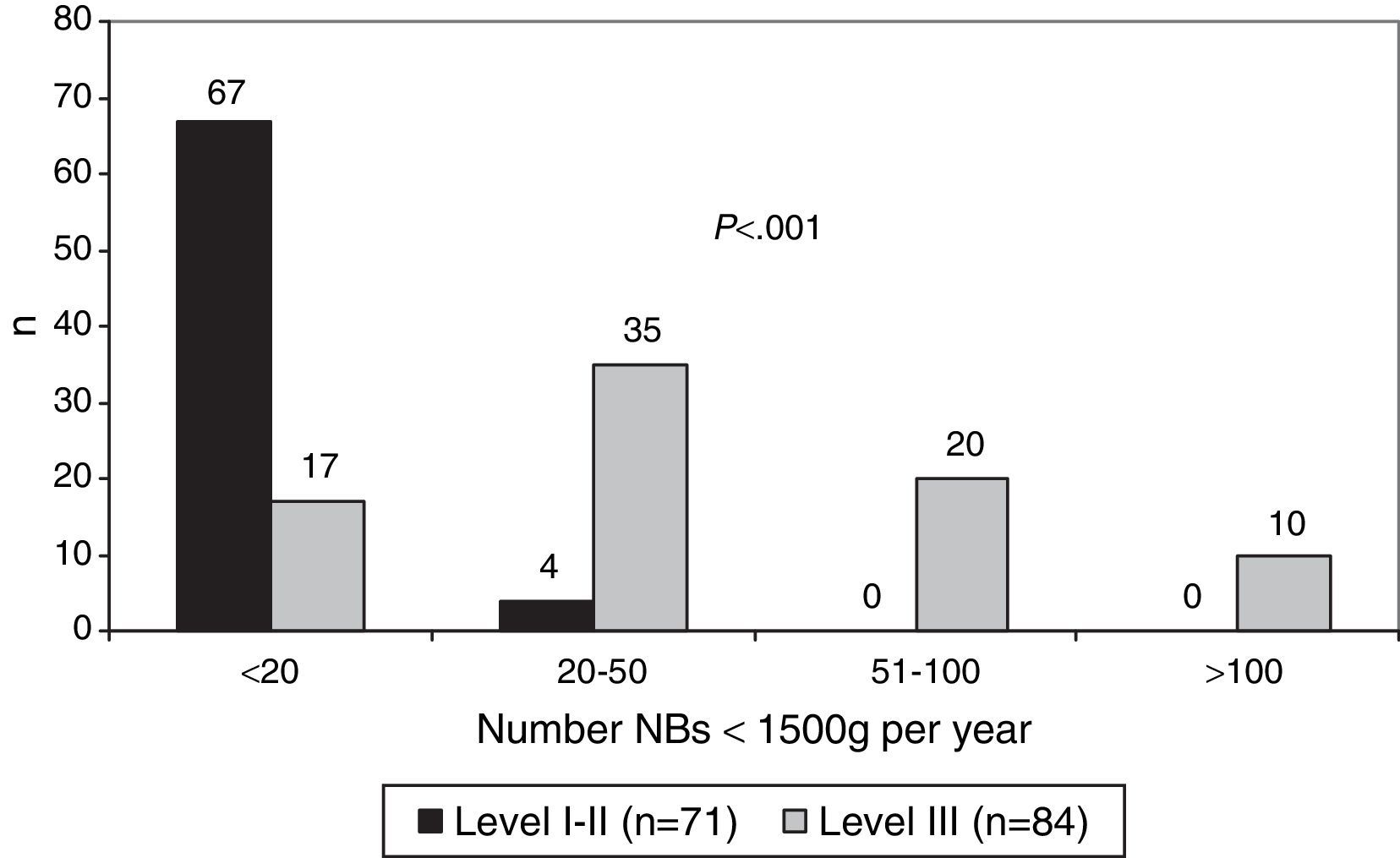
We received a total of 155 responses to the 180 questionnaires that we sent (86%). Of the 155 hospitals that participated, 35 were level I (23%), 36 level II (23%) and 84 level III (54%): 36 were level IIIA (23%), 38 level IIIB (24.5%) and 10 level IIIC (6.5%). Fig. 1 shows the number of newborns (NBs) with birth weights less than 1500g (very low birth weight) born each year in hospitals offering different levels of care.
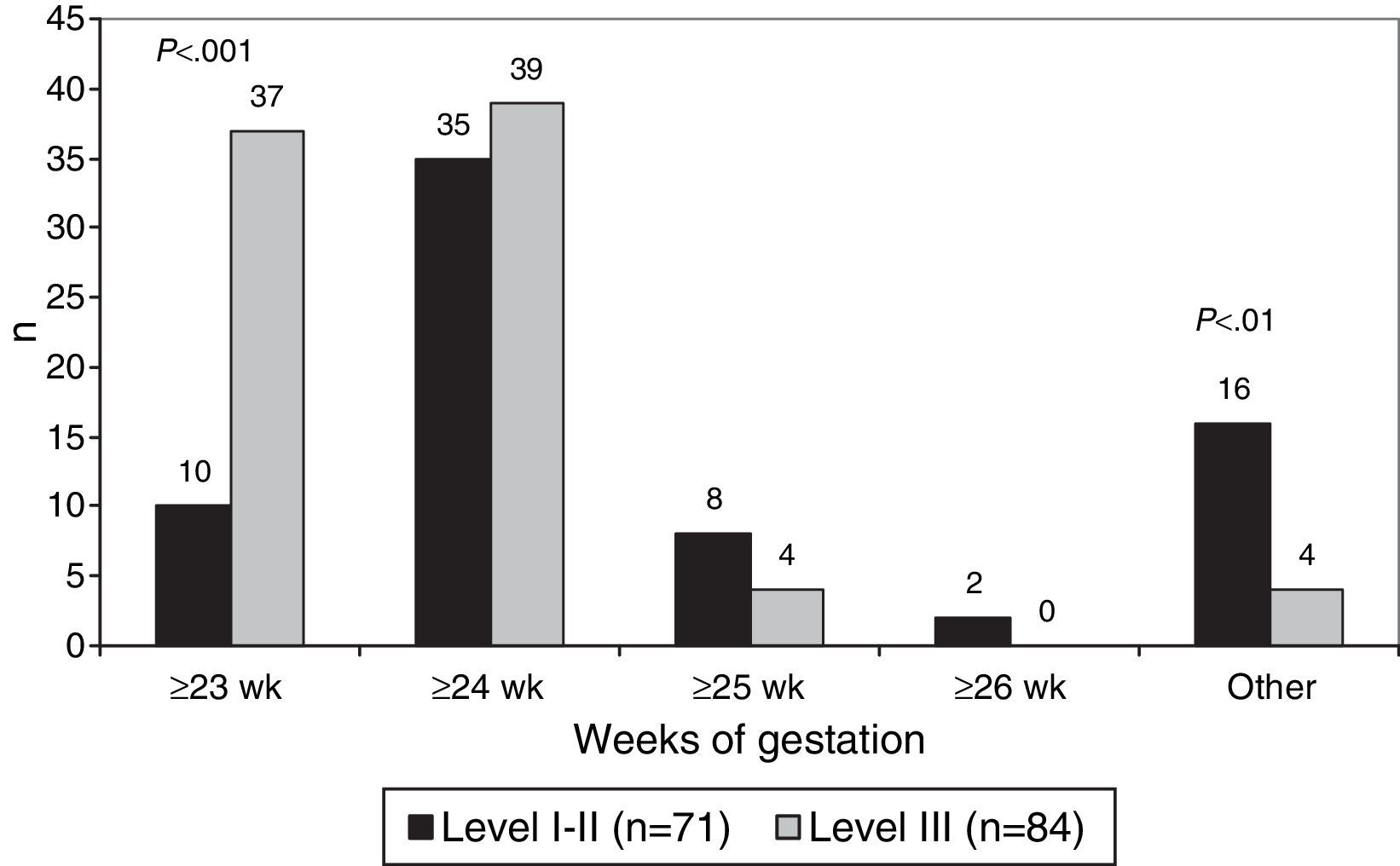
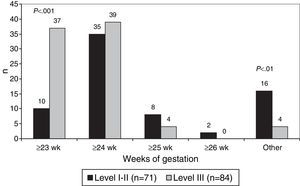
The lowest gestational age at which resuscitation was attempted was 23 weeks in 47 hospitals (30.3%), 24 weeks in 74 hospitals (47.7%) and 25 weeks in 12 hospitals (7.7%). Resuscitation at the limit of viability of 23 weeks was more frequent in level III hospitals (P<.001) (Fig. 2).
Areas equipped for neonatal resuscitation in the delivery room are arranged in the same room as the mother or in an adjacent room in 72.3% and 62.6% of hospitals, respectively.
When it came to the presence of the father or the mother's partner in the delivery room, 97.5% of hospitals offered the opportunity to accompany the mother, although only 26.5% allowed this in any type of birth, including caesarean deliveries. The birth plan submitted by the parents was accepted and adhered to in 75.5% of the hospitals, with no differences based on level of care.
The family participated in the decision to withhold or withdraw resuscitation along with the medical team in 62.0% of level I–II hospitals and 84.5% of level III hospitals (P<.01). The withdrawal of resuscitation when the patient is unresponsive (Apgar score remaining at 0) is indicated at 10, 15 and 20min in 51.6%, 28.8% and 18.3% of level I, II and III hospitals, respectively. Compared to level I and II hospitals, withdrawal of resuscitation at 10min was more frequent in level III hospitals (61.9% vs 38.0%; P<.01).
Resuscitation team composition and trainingIn level III hospitals, resuscitation teams (RTs) were mostly composed of neonatologists and paediatric residents (94.0% and 78.6%, respectively), while in level I and II hospitals, general paediatricians (93.0%), midwives (76.1%) and anaesthesiologists (64.8%) were the most frequent providers (P<.001). Two or more qualified RT members were available 24h a day in 80% of the hospitals, with a higher percentage in level III facilities (94.0% compared to 63.9%; P<.001).
Neonatal resuscitation courses were held in 90.3% of the hospitals, most often on a yearly basis (40%). Training by instructors accredited by the GRN-SENeo was more frequent in level III hospitals (94.0% vs 71.8%; P<.001), as were certification courses (97.6% vs 81.7%, P<.01). Five hospitals (all level III) performed video recordings in the delivery room, and 20.6% of the hospitals offered simulation-based courses.
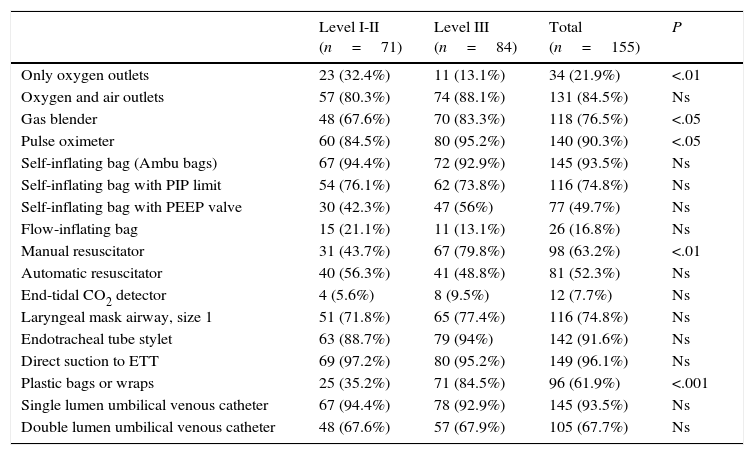
Equipment in the resuscitation areaTable 2 shows the data for resuscitation equipment. Oxygen-only outlets were more frequent in level I and II hospitals, while oxygen/air blenders, pulse oximeters, manual resuscitators and plastic wrap were predominantly available in level III hospitals.
Equipment in the resuscitation area.
| Level I-II (n=71) | Level III (n=84) | Total (n=155) | P | |
|---|---|---|---|---|
| Only oxygen outlets | 23 (32.4%) | 11 (13.1%) | 34 (21.9%) | <.01 |
| Oxygen and air outlets | 57 (80.3%) | 74 (88.1%) | 131 (84.5%) | Ns |
| Gas blender | 48 (67.6%) | 70 (83.3%) | 118 (76.5%) | <.05 |
| Pulse oximeter | 60 (84.5%) | 80 (95.2%) | 140 (90.3%) | <.05 |
| Self-inflating bag (Ambu bags) | 67 (94.4%) | 72 (92.9%) | 145 (93.5%) | Ns |
| Self-inflating bag with PIP limit | 54 (76.1%) | 62 (73.8%) | 116 (74.8%) | Ns |
| Self-inflating bag with PEEP valve | 30 (42.3%) | 47 (56%) | 77 (49.7%) | Ns |
| Flow-inflating bag | 15 (21.1%) | 11 (13.1%) | 26 (16.8%) | Ns |
| Manual resuscitator | 31 (43.7%) | 67 (79.8%) | 98 (63.2%) | <.01 |
| Automatic resuscitator | 40 (56.3%) | 41 (48.8%) | 81 (52.3%) | Ns |
| End-tidal CO2 detector | 4 (5.6%) | 8 (9.5%) | 12 (7.7%) | Ns |
| Laryngeal mask airway, size 1 | 51 (71.8%) | 65 (77.4%) | 116 (74.8%) | Ns |
| Endotracheal tube stylet | 63 (88.7%) | 79 (94%) | 142 (91.6%) | Ns |
| Direct suction to ETT | 69 (97.2%) | 80 (95.2%) | 149 (96.1%) | Ns |
| Plastic bags or wraps | 25 (35.2%) | 71 (84.5%) | 96 (61.9%) | <.001 |
| Single lumen umbilical venous catheter | 67 (94.4%) | 78 (92.9%) | 145 (93.5%) | Ns |
| Double lumen umbilical venous catheter | 48 (67.6%) | 57 (67.9%) | 105 (67.7%) | Ns |
Ns, not significant; PEEP, positive end-expiratory pressure; PIP, peak inspiratory pressure; ETT, endotracheal tube.
Tubs for water births were available in 17.4% de of the hospitals, and were more common in level I and II facilities (25.4% vs 10.7%; P<.05).
Clinical aspects of the resuscitation processThe temperature of the NB in the delivery room is “never or rarely” documented in 72.3% of the hospitals, while it is “always” measured in 14.2%, predominantly in level I and II hospitals (21.1% vs 8.3%; P<.05). The risk of hypoxic-ischaemic encephalopathy (HIE) is the clinical situation cited most frequently as a specific reason to measure the temperature of NBs in the delivery room, followed by preterm birth. The thermal measures used to prevent heat loss in preterm infants include radiant infant warmers in 100% of the hospitals, hats in 98% and warmed blankets in 71.6%. Polyethylene bags are used in 63.9% of the centres, predominantly in level III hospitals (83.3% vs 40.8%, P<.001). Only 30.3% of the hospitals maintained appropriate room temperatures (≥26°C), and the use of radiant infant warmers and hats reached 100% and 98.1%, respectively. Therapeutic hypothermia for the treatment of HIE is initiated in the delivery room in 79% of the hospitals (levels I and II, 66%; level III, 90%; P<.001), most frequently by removing the source of heat after stabilising the patient (80.6%). The temperature of the NB is “always or frequently” measured within 1h of admission to the neonatal intensive care unit (NICU) in 94% of the hospitals.
As for the use of oxygen in delivery rooms, 36% of hospitals administer free-flow oxygen (56.3% of levels I–II vs 19% of level III; P<.001). Pulse oximetry is used in 90.3% of hospitals (“always or frequently” in 78.0%). The most frequent reason cited for “rarely or never” using pulse oximetry is the response delay. In term NBs, resuscitation is initiated with room air in 89.7% of hospitals. In preterm NBs, an initial fraction of inspired oxygen (FIO2) of 0.3–0.5 is used in 73.2% of level I and II hospitals compared to 88.1% of level III hospitals (P<.05). During resuscitation, FIO2 is adjusted based on Sp02 values in 94.0% of level III hospitals and 77.5% of level I and II hospitals (P<.01), and based on the colour of the NB in 59.2% of level I and II hospitals and 31.0% of level III hospitals (P<.001). Resuscitation areas are equipped with carbon dioxide (CO2) detectors in 5.2% of the hospitals, and used to confirm correct intubation of the NB in only 2.6%.
The device used most commonly to administer positive pressure with a face mask is the “manual resuscitator” (61.9% of hospitals, with predominance of level III facilities: 78.6% vs 42.3%; P<.001).
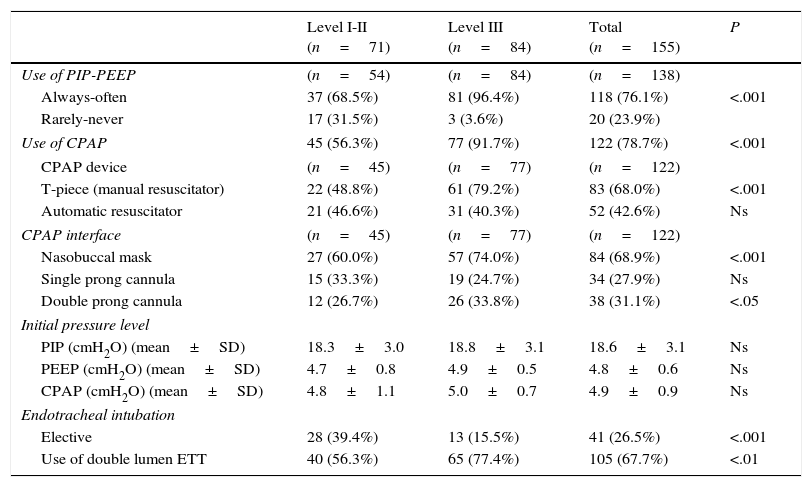
Table 3 summarises the ventilatory support practices in preterm NBs. Peak inspiratory pressure (PIP), positive end-expiratory pressure (PEEP) and continuous positive airway pressure (CPAP) are used more frequently in level III facilities. The CPAP device used most frequently is “T-piece” manual resuscitator (68%) and the most common CPAP interface is the nasobuccal mask (68.9%), used most frequently in level III hospitals (P<.001). Sustained lung inflation is used in 11% of hospitals, with no uniformity in the PIP used or the duration of inflations. Elective endotracheal intubation is used more frequently in level I and II facilities (P<.001), but the use of double lumen endotracheal tubes is more frequent in tertiary care hospitals (P<.01). Forty-seven percent of hospitals administer surfactant in the delivery room.
Respiratory care of preterm newborns in the delivery room.
| Level I-II (n=71) | Level III (n=84) | Total (n=155) | P | |
|---|---|---|---|---|
| Use of PIP-PEEP | (n=54) | (n=84) | (n=138) | |
| Always-often | 37 (68.5%) | 81 (96.4%) | 118 (76.1%) | <.001 |
| Rarely-never | 17 (31.5%) | 3 (3.6%) | 20 (23.9%) | |
| Use of CPAP | 45 (56.3%) | 77 (91.7%) | 122 (78.7%) | <.001 |
| CPAP device | (n=45) | (n=77) | (n=122) | |
| T-piece (manual resuscitator) | 22 (48.8%) | 61 (79.2%) | 83 (68.0%) | <.001 |
| Automatic resuscitator | 21 (46.6%) | 31 (40.3%) | 52 (42.6%) | Ns |
| CPAP interface | (n=45) | (n=77) | (n=122) | |
| Nasobuccal mask | 27 (60.0%) | 57 (74.0%) | 84 (68.9%) | <.001 |
| Single prong cannula | 15 (33.3%) | 19 (24.7%) | 34 (27.9%) | Ns |
| Double prong cannula | 12 (26.7%) | 26 (33.8%) | 38 (31.1%) | <.05 |
| Initial pressure level | ||||
| PIP (cmH2O) (mean±SD) | 18.3±3.0 | 18.8±3.1 | 18.6±3.1 | Ns |
| PEEP (cmH2O) (mean±SD) | 4.7±0.8 | 4.9±0.5 | 4.8±0.6 | Ns |
| CPAP (cmH2O) (mean±SD) | 4.8±1.1 | 5.0±0.7 | 4.9±0.9 | Ns |
| Endotracheal intubation | ||||
| Elective | 28 (39.4%) | 13 (15.5%) | 41 (26.5%) | <.001 |
| Use of double lumen ETT | 40 (56.3%) | 65 (77.4%) | 105 (67.7%) | <.01 |
CPAP, continuous positive airway pressure; Ns, not significant; PEEP, positive end expiratory pressure; PIP, peak inspiratory pressure; ETT, endotracheal tube.
Intratracheal adrenaline is the medication used most frequently in the delivery room (96.8%), followed by surfactant (47.1%). Intratracheal adrenaline is most commonly administered in doses ranging from 0.05 to 0.1mg/kg (65% of hospitals). Sodium bicarbonate is used “rarely” during prolonged resuscitation in 86.4% of the hospitals. Naloxone is still used in the delivery room in 33.5% of hospitals. We did not find significant differences in the use of medication based on hospital level of care.
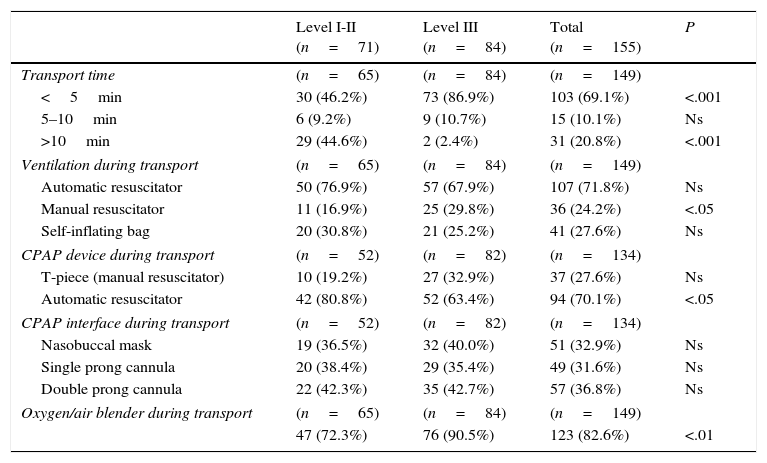
Transport of the newborn from the delivery room to the neonatal intensive care unitTable 4 summarises the data pertaining to the transport of NBs from the delivery room to the neonatal intensive care unit (NICU). Level III hospitals have shorter transport times (P<.001) and use manual resuscitators and gas blenders more frequently (P<.05 and P<.01, respectively); while level I and II hospitals use transport ventilators with CPAP more frequently (P<0.05).
Transport from delivery room to neonatal unit.
| Level I-II (n=71) | Level III (n=84) | Total (n=155) | P | |
|---|---|---|---|---|
| Transport time | (n=65) | (n=84) | (n=149) | |
| <5min | 30 (46.2%) | 73 (86.9%) | 103 (69.1%) | <.001 |
| 5–10min | 6 (9.2%) | 9 (10.7%) | 15 (10.1%) | Ns |
| >10min | 29 (44.6%) | 2 (2.4%) | 31 (20.8%) | <.001 |
| Ventilation during transport | (n=65) | (n=84) | (n=149) | |
| Automatic resuscitator | 50 (76.9%) | 57 (67.9%) | 107 (71.8%) | Ns |
| Manual resuscitator | 11 (16.9%) | 25 (29.8%) | 36 (24.2%) | <.05 |
| Self-inflating bag | 20 (30.8%) | 21 (25.2%) | 41 (27.6%) | Ns |
| CPAP device during transport | (n=52) | (n=82) | (n=134) | |
| T-piece (manual resuscitator) | 10 (19.2%) | 27 (32.9%) | 37 (27.6%) | Ns |
| Automatic resuscitator | 42 (80.8%) | 52 (63.4%) | 94 (70.1%) | <.05 |
| CPAP interface during transport | (n=52) | (n=82) | (n=134) | |
| Nasobuccal mask | 19 (36.5%) | 32 (40.0%) | 51 (32.9%) | Ns |
| Single prong cannula | 20 (38.4%) | 29 (35.4%) | 49 (31.6%) | Ns |
| Double prong cannula | 22 (42.3%) | 35 (42.7%) | 57 (36.8%) | Ns |
| Oxygen/air blender during transport | (n=65) | (n=84) | (n=149) | |
| 47 (72.3%) | 76 (90.5%) | 123 (82.6%) | <.01 | |
CPAP, continuous positive airway pressure; Ns, not significant.
This is the largest survey on neonatal resuscitation ever carried out in Spain, and the second published by the GRN-SENeo since its inception. In 2011 there were 470553 live births in Spain15; of which 347210 were attended at hospitals that participated in this survey, amounting to approximately 74% of the total births in that year. Compared to the previous survey,14 43 more hospitals participated in this one, representing 14% more of the total annual number of births. We have estimated that 4900 very low birth weight NBs receive care in the participating centres each year, a figure far exceeding the 2700 in the SEN 1500 morbidity and mortality report for year 2011.16 These figures demonstrate that the results of the survey are representative of health care practices in Spain.
The definition of the limits of viability in preterm NBs varies from country to country, and in Spain, the GRN-SENeo recommends initiating resuscitation in newborns at the threshold of 23 or 24 weeks of gestation (WG) on a case-to-case basis, taking into consideration the wishes of the parents and the morbidity and mortality rates of the particular hospital.6 According to our data, 78% of participating hospitals initiate reanimation at 23–24WG. This finding stands in contrast with data from other countries such as Japan or Germany, which have set thresholds at 22–23 WG.17,18
There is no consensus on when to discontinue resuscitation efforts when there is no detectable heart rate. Half of the participating hospitals discontinue at 10min from birth, with predominance of level III hospitals. Although the ILCOR2 considers that it is appropriate to discontinue resuscitation if there has been no detectable heart rate for 10min, and that more data are required to establish recommendations for cases of persistent bradycardia (HR<60bpm) 10–15min after birth, in actual clinical practice it may be difficult to determine when resuscitation should be discontinued. Follow-up studies in the age of therapeutic hypothermia challenge the validity of this stance, as one out of five NB with an Apgar score of 0/0/0 have no moderate or severe sequelae at ages 18–22 months19 and 6–7 years.20 One editorial has addressed this issue and concluded that we should use the data available for the age of hypothermia to reflect on the optimal moment for deciding whether to maintain or withdraw life support in NBs with very low Apgar scores. Some authors affirm that delaying this decision would allow a more accurate prognosis and enough time to explore the wishes of the parents.21
The Neonatal Resuscitation Program (NRP) of the American Academy of Pediatrics recommends that at least two members of the resuscitation team be physically present around the clock to provide care for at-risk NBs in the delivery room.22 In Spain, 94% of level III hospitals have at least two members available 24h a day, a percentage that has remained stable in the past five years7; team members are most frequently neonatologists in level III hospitals (94%) and general paediatricians in level I and II hospitals (93%). A survey conducted in the United States8 showed that 31% of resuscitation teams usually consisted of two members, so we find our data encouraging in this regard.
Multiple studies demonstrate the benefits of neonatal resuscitation programmes in the training of professionals that provide neonatal care at birth, in developed as well as developing and emerging countries.22–24 In addition to improving the knowledge, skills and efficiency of providers,24 such training can have a positive effect on various indicators (Apgar score, length of stay and neonatal mortality).22,23 In 2011, there were 5304 health professionals that had completed the Neonatal Reanimation courses accredited by the GRN-SENeo, and 392 active instructors.6 Data from this survey showed that the courses were offered in more than 90% of the hospitals or their service areas (97% for level III hospitals), while 84% of the participating hospitals had instructors accredited by the GRN-SENeo (94% of level III hospitals), figures that have experienced a sustained increase in recent years. These courses are based on simulation techniques and employ group dynamics, distraction, booster practice and integrated feedback, an approach proven to be highly effective.25
The equipment available in the delivery room is a good indicator of neonatal resuscitation practices. A Japanese survey26 found variability in the equipment available in different developed countries. In Spain, we found a greater availability of some equipment in level III hospitals (Table 2).
The 2010 recommendations stated that the use of CO2 detectors combined with clinical assessment is the most reliable method to confirm tracheal tube placement in neonates with cardiac output.2,4,27 One aspect deserving consideration and that could be improved in the future is the low availability of CO2 detectors in Spanish delivery rooms (7.7%), which is inferior to that of other countries, such as the United States (32%),8 Austria (36%),10 Japan (45%)26 or Canada (87%).13
Water births are an alternative to conventional delivery that is becoming increasingly popular in many countries.28 In Spain, 17.4% of hospitals have tubs available to attend labour and delivery in water, an availability that is greater in level I and II hospitals, a fact that would explain the hospitals’ choice to offer this modality. The literature has reported that at least 143 maternity units in the United States29 and 295 in the United Kingdom30 have tubs.
Therapeutic hypothermia is an accepted clinical practice in term and near-term NBs with moderate or severe HIE within 6h of birth.2–4 In Spain, passive hypothermia is initiated in the delivery room in 79.4% of hospitals (90.5% of level III hospitals), and the most frequent measure consists of switching off the radiant warmer once the NB is stable (80.6%).
Initiation of resuscitation with room air in term NBs is considered appropriate by the 2010 guidelines2–4 based on studies that demonstrated that the use of oxygen at high concentrations leads to less favourable outcomes and offers no benefits compared to the use of air.31–33 With the purpose of contributing to the rational use of oxygen, several algorithms2–6 use variations of normal pulse oximeter parameters,34 including SpO2 targets, during the first 10min of resuscitation.
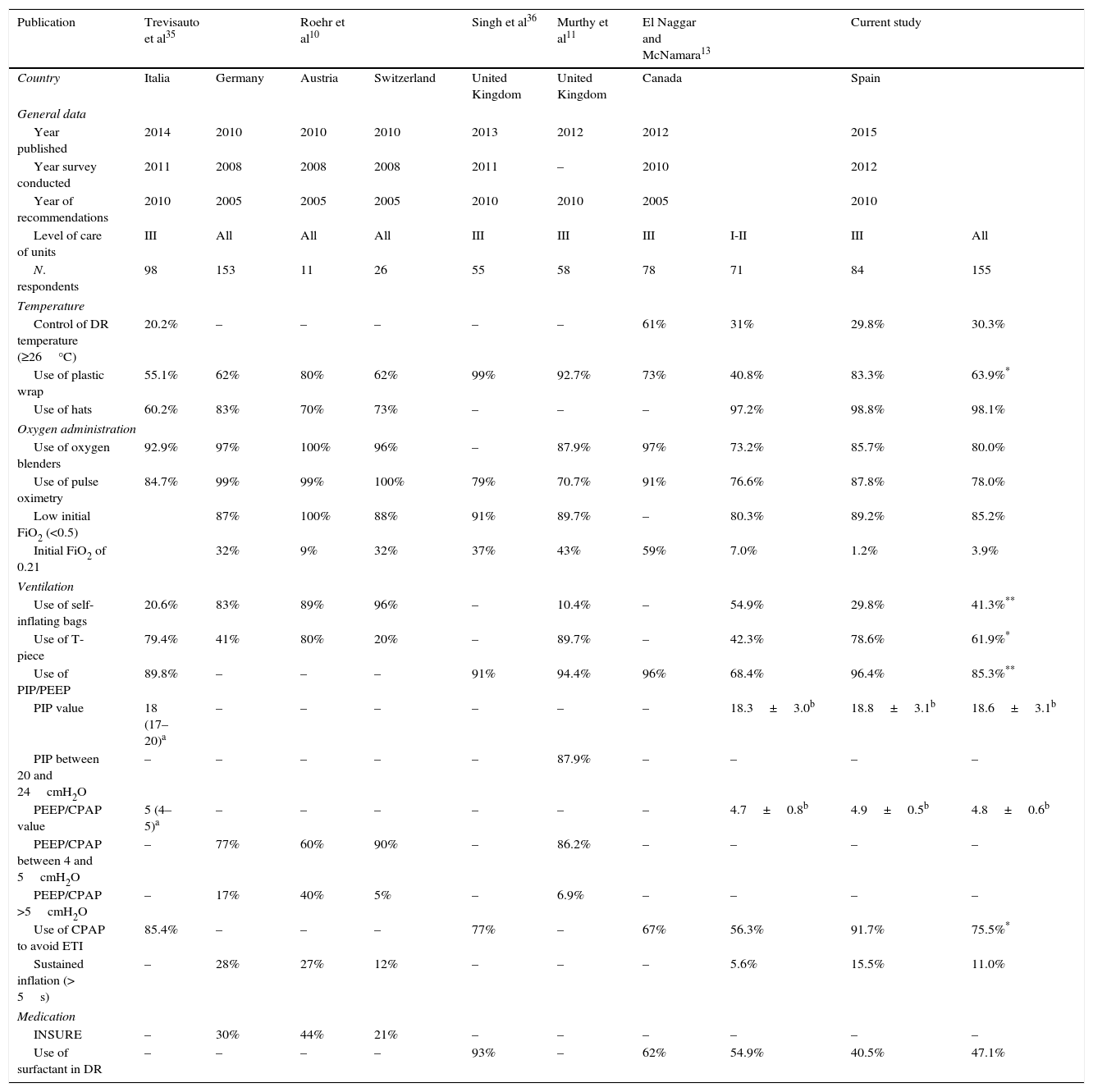
Table 5 presents the clinical practices in the resuscitation of preterm NBs in different countries.10,11,13,35,36 Several measures are used to maintain body temperature in NBs less than 28 weeks of gestational age, including novel ones like the use of plastic wrap and an ambient temperature of at least 26°C in the delivery room.2,37 In Spanish hospitals, the use of radiant warmers and hats is nearly universal, while high room ambient temperatures (30%) and the use of polyethylene wrap could improve. In most countries, the use of plastic wrap ranges between 55% and 80%, although it is currently higher in the United Kingdom. The initial FiO2 for use in preterm NBs has not been established,2 and most countries, Spain included, use O2 concentrations of less than 50%. When it comes to ventilation in preterm NBs, there is variability in the devices used, with predominance of PIP and PEEP control (T-piece) and CPAP with the aim of avoiding tracheal intubation. There pressure limits used (PIP, PEEP and CPAP) are increasingly uniform. At present, sustained inflation is not a widespread practice.
Summary of surveys regarding equipment and clinical practices in the resuscitation of preterm newborns in the delivery room.
| Publication | Trevisauto et al35 | Roehr et al10 | Singh et al36 | Murthy et al11 | El Naggar and McNamara13 | Current study | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Country | Italia | Germany | Austria | Switzerland | United Kingdom | United Kingdom | Canada | Spain | ||
| General data | ||||||||||
| Year published | 2014 | 2010 | 2010 | 2010 | 2013 | 2012 | 2012 | 2015 | ||
| Year survey conducted | 2011 | 2008 | 2008 | 2008 | 2011 | – | 2010 | 2012 | ||
| Year of recommendations | 2010 | 2005 | 2005 | 2005 | 2010 | 2010 | 2005 | 2010 | ||
| Level of care of units | III | All | All | All | III | III | III | I-II | III | All |
| N. respondents | 98 | 153 | 11 | 26 | 55 | 58 | 78 | 71 | 84 | 155 |
| Temperature | ||||||||||
| Control of DR temperature (≥26°C) | 20.2% | – | – | – | – | – | 61% | 31% | 29.8% | 30.3% |
| Use of plastic wrap | 55.1% | 62% | 80% | 62% | 99% | 92.7% | 73% | 40.8% | 83.3% | 63.9%* |
| Use of hats | 60.2% | 83% | 70% | 73% | – | – | – | 97.2% | 98.8% | 98.1% |
| Oxygen administration | ||||||||||
| Use of oxygen blenders | 92.9% | 97% | 100% | 96% | – | 87.9% | 97% | 73.2% | 85.7% | 80.0% |
| Use of pulse oximetry | 84.7% | 99% | 99% | 100% | 79% | 70.7% | 91% | 76.6% | 87.8% | 78.0% |
| Low initial FiO2 (<0.5) | 87% | 100% | 88% | 91% | 89.7% | – | 80.3% | 89.2% | 85.2% | |
| Initial FiO2 of 0.21 | 32% | 9% | 32% | 37% | 43% | 59% | 7.0% | 1.2% | 3.9% | |
| Ventilation | ||||||||||
| Use of self-inflating bags | 20.6% | 83% | 89% | 96% | – | 10.4% | – | 54.9% | 29.8% | 41.3%** |
| Use of T-piece | 79.4% | 41% | 80% | 20% | – | 89.7% | – | 42.3% | 78.6% | 61.9%* |
| Use of PIP/PEEP | 89.8% | – | – | – | 91% | 94.4% | 96% | 68.4% | 96.4% | 85.3%** |
| PIP value | 18 (17–20)a | – | – | – | – | – | – | 18.3±3.0b | 18.8±3.1b | 18.6±3.1b |
| PIP between 20 and 24cmH2O | – | – | – | – | – | 87.9% | – | – | – | – |
| PEEP/CPAP value | 5 (4–5)a | – | – | – | – | – | – | 4.7±0.8b | 4.9±0.5b | 4.8±0.6b |
| PEEP/CPAP between 4 and 5cmH2O | – | 77% | 60% | 90% | – | 86.2% | – | – | – | – |
| PEEP/CPAP >5cmH2O | – | 17% | 40% | 5% | – | 6.9% | – | – | – | – |
| Use of CPAP to avoid ETI | 85.4% | – | – | – | 77% | – | 67% | 56.3% | 91.7% | 75.5%* |
| Sustained inflation (> 5s) | – | 28% | 27% | 12% | – | – | – | 5.6% | 15.5% | 11.0% |
| Medication | ||||||||||
| INSURE | – | 30% | 44% | 21% | – | – | – | – | – | – |
| Use of surfactant in DR | – | – | – | – | 93% | – | 62% | 54.9% | 40.5% | 47.1% |
CPAP, continuous positive airway pressure; FiO2, fraction of inspired oxygen; INSURE, intubation-surfactant administration-early extubation; PEEP, positive end-expiratory pressure; PIP, peak inspiratory pressure; DR, delivery room; ETI, endotracheal intubation.
When it comes to the transport of the NB from the delivery room to the NICU, the most commonly used ventilation systems were automatic resuscitators, and we have observed an increase in the use of CPAP and oxygen blenders in Spanish hospitals.
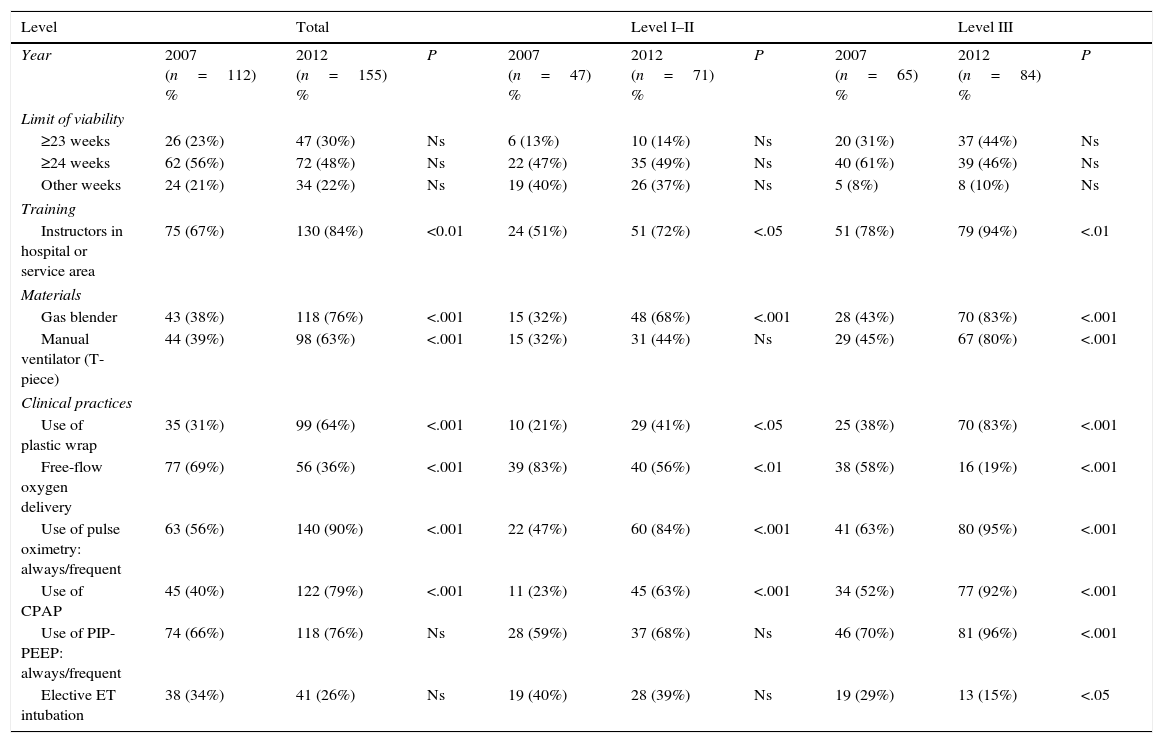
Table 6 compares the data on resuscitation equipment and practices of the latest survey on neonatal resuscitation in Spain7 and the current study. Between 2007 and 2012 there has been an increase in the availability of gas blenders and manual resuscitators (T-pieces) and in the use of pulse oximetry for the adjustment of FiO2, and a decrease in the administration of free-flow oxygen. We also found evidence of increased use of pressure-controlled devices in level III units and early CPAP to facilitate alveolar recruitment.
Comparison of the data from the 2007a and 2012 (current study) SENeo surveys.
| Level | Total | Level I–II | Level III | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Year | 2007 (n=112) % | 2012 (n=155) % | P | 2007 (n=47) % | 2012 (n=71) % | P | 2007 (n=65) % | 2012 (n=84) % | P |
| Limit of viability | |||||||||
| ≥23 weeks | 26 (23%) | 47 (30%) | Ns | 6 (13%) | 10 (14%) | Ns | 20 (31%) | 37 (44%) | Ns |
| ≥24 weeks | 62 (56%) | 72 (48%) | Ns | 22 (47%) | 35 (49%) | Ns | 40 (61%) | 39 (46%) | Ns |
| Other weeks | 24 (21%) | 34 (22%) | Ns | 19 (40%) | 26 (37%) | Ns | 5 (8%) | 8 (10%) | Ns |
| Training | |||||||||
| Instructors in hospital or service area | 75 (67%) | 130 (84%) | <0.01 | 24 (51%) | 51 (72%) | <.05 | 51 (78%) | 79 (94%) | <.01 |
| Materials | |||||||||
| Gas blender | 43 (38%) | 118 (76%) | <.001 | 15 (32%) | 48 (68%) | <.001 | 28 (43%) | 70 (83%) | <.001 |
| Manual ventilator (T-piece) | 44 (39%) | 98 (63%) | <.001 | 15 (32%) | 31 (44%) | Ns | 29 (45%) | 67 (80%) | <.001 |
| Clinical practices | |||||||||
| Use of plastic wrap | 35 (31%) | 99 (64%) | <.001 | 10 (21%) | 29 (41%) | <.05 | 25 (38%) | 70 (83%) | <.001 |
| Free-flow oxygen delivery | 77 (69%) | 56 (36%) | <.001 | 39 (83%) | 40 (56%) | <.01 | 38 (58%) | 16 (19%) | <.001 |
| Use of pulse oximetry: always/frequent | 63 (56%) | 140 (90%) | <.001 | 22 (47%) | 60 (84%) | <.001 | 41 (63%) | 80 (95%) | <.001 |
| Use of CPAP | 45 (40%) | 122 (79%) | <.001 | 11 (23%) | 45 (63%) | <.001 | 34 (52%) | 77 (92%) | <.001 |
| Use of PIP-PEEP: always/frequent | 74 (66%) | 118 (76%) | Ns | 28 (59%) | 37 (68%) | Ns | 46 (70%) | 81 (96%) | <.001 |
| Elective ET intubation | 38 (34%) | 41 (26%) | Ns | 19 (40%) | 28 (39%) | Ns | 19 (29%) | 13 (15%) | <.05 |
CPAP, continuous positive airway pressure; ET, endotracheal; Ns, not significant; PEEP, positive end expiratory pressure; PIP, peak inspiratory pressure.
To conclude, we want to highlight the improvement in the neonatal resuscitation equipment and practices in Spanish delivery rooms. Some of the areas for improvement are minimising the differences between different-level hospitals, maintaining higher ambient temperatures, and increasing the use of plastic wrap, gas blenders, pulse oximetry and CO2 detectors. This survey may be useful for planning the equipment of delivery rooms and improving health care practices.
Conflicts of interestThe authors have no conflicts of interest to declare.
The GRN-SENeo thanks all the neonatologists and paediatricians that kindly agreed to participate in this study in representation of the following hospitals: Hospital Belén (A Coruña); Hospital Juan Canalejo (A Coruña); Complejo Hospitalario Universitario de Albacete (Albacete); Hospital Príncipe de Asturias (Alcalá de Henares); Hospital Comarcal de Alcoy (Alcoy); Hospital San Juan (Alicante); Hospital General Universitario de Alicante (Alicante); Hospital General de Almansa (Almansa); Complejo Hospitalario Torrecárdenas (Almería); Hospital de Aranda de Duero (Burgos); Hospital del Tajo (Aranjuez); Hospital Sanitas La Zarzuela (Aravaca); Hospital del Sureste (Arganda del Rey); Hospital del Oriente de Asturias (Arriondas); Hospital Nuestra Señora de Sonsoles (Ávila); Hospital de San Agustín (Ávilés); Hospital Universitari Germans Trias i Pujol (Badalona); Hospital Universitario de Cruces (Barakaldo, Bilbao); Hospital Universitari Sant Joan de Déu (Barcelona); Hospital Universitari Vall d’Hebron (Barcelona); Hospital Barcelona (SCIAS) (Barcelona); Hospital de la Santa Creu i Sant Pau (Barcelona); Hospital Clínic Seu Maternitat (Barcelona); Hospital Universitari Quirón Dexeus (Barcelona); Hospital Quirón (Barcelona); Clínica Corachan (Barcelona); Hospital del Mar (Barcelona); Clínica del Pilar (Barcelona); Clínica Delfos (Barcelona); Hospital Quirón Teknon (Barcelona); Hospital Universitario de Basurto (Bilbao); Hospital Universitario Montepríncipe (Boadilla del Monte, Madrid); Hospital Da Costa (Burela); Hospital Universitario (Burgos); Hospital La Mancha Centro (Ciudad Real); Hospital Infanta Margarita (Cabra); Hospital San Pedro de Alcántara (Cáceres); Hospital Universitario Puerta del Mar (Cádiz); Fundación Hospital Calahorra (Calahorra); Hospital Sant Jaume (Calella) Hospital General Universitario Santa Lucía (Cartagena); Hospital General Universitari de Castelló (Castellón); Hospital General Universitario Ciudad Real (Ciudad Real); Hospital Universitario Reina Sofía (Córdoba); Hospital del Vendrell (El Vendrell); Hospital General de Elche (Elche); Hospital de Elda Virgen de la Salud (Elda); Hospital García Orcoyen (Estella); Complejo Universitario Marcide-Profesor Novoa Santos (Ferrol); Hospital de Figueres (Figueres); Hospital Universitario de Fuenlabrada (Fuenlabrada); Hospital Universitario de Getafe (Getafe); Hospital de Cabueñes (Gijón); Hospital Universitari Dr. Josep Trueta (Girona); Clínica Girona (Girona); Hospital Universitario Virgen de las Nieves (Granada); Hospital General de Granollers (Granollers); Hospital General de l’Hospitalet (L’Hospitalet de Llobregat); Hospital Juan Ramón Jiménez (Huelva); Hospital Can Misses (Ibiza); Hospital General d’Igualada (Igualada); Complejo Hospitalario de Jaén (Jaén); Hospital Universitario de Canarias (La Laguna, Tenerife); Hospital SAS de la Línea de la Concepción (La Línea, Cádiz); Fundació Sant Hospital de La Seu d’Urgell (La Seu d’Urgell); Hospital Universitario Insular de Gran Canaria (Las Palmas de Gran Canaria); Hospital Universitario Severo-Ochoa (Leganés); Hospital de León (León); Hospital Universitari Arnau de Vilanova (Lleida); Hospital San Pedro (Logroño); Hospital Universitario 12 de Octubre (Madrid); Hospital Universitario Gregorio Marañón (Madrid); Hospital Clínico San Carlos (Madrid); Hospital Universitario La Paz (Madrid); Hospital Capio Alcorcón Sur (Madrid); Hospital Nuevo Belén (Madrid); Hospital Universitario Infanta Leonor (Madrid); Hospital Infanta Elena (Madrid); Hospital Universitario Puerta de Hierro (Mahadahonda. Madrid); Hospital Carlos Haya (Málaga); Hospital de Manresa (Manresa); Hospital Costa del Sol (Marbella); Hospital Sant Joan de Déu (Martorell); Hospital de Medina del Campo (Medina del Campo); Hospital de Mendaro (Mendaro, Guipúzcoa); Hospital Mateu Orfila (Menorca); Hospital de Mérida (Mérida); Hospital General de Riotinto (Minas de Riotinto); Hospital de Mollet (Mollet); Hospital Alto Deba (Mondragón-Arrasate); Hospital de Monforte (Monforte de Lemos); Hospital de Montilla (Montilla); Hospital Comarcal de Mora d’Ebre (Mora d’Ebre); Hospital Universitario de Móstoles (Móstoles); Hospital Rey Juan Carlos (Móstoles); Hospital Clínico Universitario Virgen de la Arrixaca (Murcia); Hospital de Sant Jaume (Olot); Hospital General d’Ontinyent (Ontinyent); Hospital de Orihuela (Orihuela); Hospital Comarcal de Osuna (Osuna); Hospital Universitario Central de Asturias (Oviedo); Hospital de Palamós (Palamós); Hospital Universitari Son Espases (Palma de Mallorca); Hospital de Son Llàtzer (Palma de Mallorca); Hospital Quirón Palma Planas (Palma de Mallorca); Hospital de Pamplona (Pamplona); Clínica Universidad de Navarra (Pamplona); Hospital del Bierzo (Ponferrada); Complejo Hospitalario de Pontevedra (Pontevedra); Hospital Valle de Los Pedroches (Pozoblanco); Fundació Hospital de Puigcerdà (Puigcerdà); Hospital Sant Joan de Reus (Reus); Hospital do Barbanza (Ribeira); Hospital General de Catalunya (Sant Cugat del Vallés); Corporació Sanitària Parc Taulí (Sabadell); Hospital de Sagunto (Sagunto); Complejo Asistencial Universitario de Salamanca (Salamanca); Hospital Santa Caterina (Salt); Hospital El Escorial (San Lorenzo del Escorial); Hospital Universitario Donostia (San Sebastián); Policlínica Guipúzcoa (San Sebastián); Parc Sanitari Sant Joan de Déu (Sant Boi de Llobregat); Hospital de l’Espèrit Sant (Santa Coloma de Gramanet); Hospital Universitario Marqués de Valdecilla (Santander); Hospital Clínico Universitario (Santiago de Compostela); Hospital Universitario Virgen del Rocío (Sevilla); Hospital Universitario Virgen Macarena (Sevilla); Hospital Universitario de Valme (Sevilla); Hospital Santa Bárbara (Soria); Hospital Universitari Joan XXIII (Tarragona); Hospital Mútua de Terrassa (Terrasa); Hospital Virgen de la Salud (Toledo); Hospital Universitario de Torrejón (Torrejon de Ardoz); Hospital Universitario HM Torrelodones (Torrelodones); Hospital Verge de la Cinta (Tortosa); Hospital Comarcal del Pallars (Tremp); Hospital San Juan de la Cruz (Úbeda); Hospital Universitario Infanta Elena (Valdemoro); Hospital Universitari Doctor Peset (Valencia); Hospital Universitari La Fe (Valencia); Hospital Quirón Valencia (Valencia); Hospital del Río Hortega (Valladolid); Hospital Clínico Universitario de Valladolid (Valladolid); Consorci Hospitalari de Vic (Vic); Complexo Hospitalario Universitario de Vigo H-Xeral (Vigo); Hospital Nuestra Señora de Fátima (Vigo); Hospital Comarcal Alt Penedès (Vilafranca del Penedès); Hospital do Salnes (Vilagarcía de Arousa); Hospital de La Plana (Vila-Real); Hospital Comarcal de Vinaros (Vinaroz); Hospital Vitoria (Vitoria); Complejo Asistencial de Zamora (Zamora); Hospital Universitario Miguel Servet (Zaragoza); Hospital Clínico Universitario Lozano Blesa (Zaragoza); Hospital de Zumarraga (Zumárraga).
Elena Burón (Hospital Clínico Universitario de Valladolid), Juana Guzmán (Hospital Universitario Reina Sofía de Córdoba), Eva González (Hospital Álvaro Cunqueiro de la Estructura Organizada de Xestión Integrada de Vigo), César Ruiz (Hospital Vall d’Hebron de Barcelona), Dolores Elorza (Hospital Universitario La Paz de Madrid), Gonzalo Zeballos (Hospital Universitario Gregorio Marañón de Madrid), Miguel Sánchez (Hospital Universitario Puerta de Hierro de Mahadahonda, Madrid).
Group members Neonatal Resuscitation of the Spanish Society of Neonatology (GRN-SENeo) are presented in Annex 1.
Please cite this article as: Iriondo M, Izquierdo M, Salguero E, Aguayo J, Vento M, Thió M. Encuesta española de reanimación neonatal 5 años después. ¿Vamos mejorando? An Pediatr (Barc). 2016;84:260–270.