Therapeutic hypothermia is the standard treatment for hypoxic ischaemic encephalopathy (HIE), despite not knowing all its effects and complications. Sinus bradycardia is one of the consequences of cooling that has been previously documented in the literature, but little is known about the cardiac electrical activity in these patients.
ObjectiveTo determine the corrected QT (QTc) interval in newborns treated with therapeutic hypothermia for HIE.
Materials and methodsA prospective observational study was conducted in all patients treated with hypothermia for HIE that were admitted to our Unit between November 2012 and October 2013. ECGs were performed during hypothermia (every 24h), during the re-warming period (at 34.5°C, 35.5°C, 36.5°C), and on the 7th day of life.
ResultsA total of 19 patients were included. A prolonged QTc was observed in all patients during hypothermia, and 84% (n=16) had prolonged QTc in all the ECGs during treatment. In 3 patients, one of the ECGs did not have a prolonged QTc. After re-warming, the QTc interval returned to normal in all patients. No statistically significant differences were seen when the degree of HIE (P=.192) or the use of inotropic support (P=.669) were considered.
ConclusionsTherapeutic hypothermia applied to asphyxiated newborns with HIE seems to induce a QTc prolongation that resolves when the patient regains physiological temperature.
La hipotermia es el tratamiento estándar en la encefalopatía hipóxico isquémica (EHI) a pesar de que todavía no se conocen todos sus efectos y complicaciones. La bradicardia sinusal está ampliamente descrita en la literatura como consecuencia de dicho enfriamiento pero se ha estudiado poco la actividad eléctrica cardiaca en este grupo de pacientes.
ObjetivoDeterminar si existe un alargamiento del intervalo QT corregido (QTc) durante la hipotermia en neonatos con EHI moderada-grave y su evolución durante el tratamiento.
Material y métodosSe reclutó a pacientes con EHI tratados con hipotermia entre noviembre del 2012 y octubre del 2013. Se realizaron electrocardiogramas (ECG) durante la hipotermia (uno cada 24 h durante el tratamiento) y durante el recalentamiento (a 34,5, 35,5 y 36,5°C). Se realizó un séptimo ECG a la semana de vida.
ResultadosSe incluyó a un total de 19 pacientes. Se observó un alargamiento del QTc en todos los pacientes durante la hipotermia. En 84% de los pacientes (n=16), el alargamiento se apreció en todos los ECG durante la hipotermia. Tras el recalentamiento, todos los pacientes presentaron una normalización del mismo. No se encontraron diferencias estadísticamente significativas al evaluar la prolongación del QTc según el grado de EHI (p=0,192) y según el uso de soporte inotrópico o no (p=0,669). Ningún paciente presentó arritmias potencialmente graves.
ConclusionesLa hipotermia moderada aplicada a los recién nacidos con EHI moderada-grave parece inducir una prolongación en el intervalo QTc temporal que se resuelve con el recalentamiento del paciente hasta temperatura fisiológica.
Moderate and severe HIE (clinically significant HIE) is an important cause of neonatal mortality and future neurodevelopmental disability. In recent years, several clinical trials and meta-analyses have demonstrated that the reduction of body temperature by 3–4°C (moderate hypothermia) by total body or selective head cooling initiated early (before 6h from birth) and maintained for 72h is effective in reducing mortality and severe disability in children that survive a perinatal hypoxic-ischaemic insult.1–4
None of the studies published to date has described significant cardiovascular or haemodynamic complications resulting from moderate hypothermia, including the development of arrhythmias, whose incidence (with the exception of sinus bradycardia) has not been observed to increase during therapeutic hypothermia,3–6 even when there has been accidental overcooling.
There is extensive documentation on the effects of hypothermia on the cardiovascular system of adults. From the earliest assessments of the effects of cooling performed in survivors of severe accidental hypothermia to animal model studies, there has been growing evidence on the association between cooling and sinus bradycardia, as well as other disturbances of the electrical activity of the heart.7–9 The joint guidelines of the International Liaison European Resuscitation Council and European Resuscitation Council10 and the guideline of the American Heart Association11 both describe the potential arrythmogenicity of hypothermia12–15 in adults treated with hypothermia after experiencing cardiac arrest due to ventricular arrhythmia.
Hypothermia has the potential to increase the risk of arrhythmia, as it decreases the rate of depolarization in pacemaker cells while simultaneously increasing the duration of action potentials. This leads to a reduction in heart rate and other abnormalities, including the prolongation of PR and QTc intervals.16,17 A prolonged QTc interval in and of itself increases the risk of developing polymorphic ventricular tachycardias.18 The evidence that is currently available is practically limited to studies conducted in adults, which have not found an increased incidence of potentially severe arrhythmias as long as the hypothermia was moderate (32–34.5°C) and electrolyte levels were monitored. There have been reports of ventricular arrhythmias in patients cooled to temperatures of less than 32°C19 and hypokalaemia.20
The aim of our study was to determine whether therapeutic hypothermia in newborns with significant EHI is associated with the incidence of arrhythmias and specifically with QTc prolongation.
Materials and methodsWe conducted an observational prospective study in newborns with neonatal asphyxia treated with moderate hypothermia that were admitted to the neonatal intensive care unit (NICU) of the Hospital Universitario Vall d’Hebron (HUVH) between November 2012 and October 2013. The study included all newborns with perinatal asphyxia and significant HIE that met the criteria for treatment with therapeutic hypothermia and received this treatment at the NICU of the HUVH during the period under study. Moderate hypothermia in all included patients consisted of total body cooling by means of a servo-controlled temperature regulation system following the protocol of the hospital.
We collected data for demographic and clinical variables of interest for the included patients (Table 1). In all patients, 12-lead electrocardiograms (ECGs) were performed during therapeutic hypothermia (one ECG every 24h during the three days of treatment) and during rewarming until the patient reached a normal body temperature (one ECG performed for every one-degree increase in body temperature: 34.5, 35.5 and 36.5°C). A seventh ECG was performed at one week of life. The ECGs were evaluated following a standardised approach by two researchers. The duration of the QTc interval was calculated by applying Bazett's formula to five cycles from the lead II tracing in each of the ECGs, and other relevant features were also assessed, including the PR interval, QRS, repolarization, heart axis and heart rate. We analysed the presence of heart rhythm and conduction abnormalities in all ECGs.
Demographic, clinical and outcome data of the patients.
| Patient | Weight (g) | GA (weeks+days) | Sex | Sentinel event | Point of origin | Degree of HIE | MRI | Seizures | Inotropic support | Death |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 3450 | 40+6 | F | Yes | VH | Severe | 8 | Yes | Yes | Yes |
| 2 | 3000 | 43+1 | F | No | VH | Moderate | 1 | Yes | Yes | No |
| 3 | 3400 | 40 | F | No | External | Moderate | 1 | No | Yes | No |
| 4 | 3020 | 38 | F | Yes | External | Moderate | 9 | Yes | Yes | Yes |
| 5 | 2400 | 40+5 | F | No | External | Moderate | 3 | No | No | No |
| 6 | 3010 | 40+2 | M | No | VH | Moderate | 0 | No | Yes | No |
| 7 | 4490 | 36+6 | M | Yes | External | Severe | 8 | Yes | No | Yes |
| 8 | 2030 | 35 | F | No | VH | Moderate | 6 | Yes | No | No |
| 9 | 2085 | 35+6 | F | No | External | Severe | 11 | Yes | No | Yes |
| 10 | 2100 | 40+4 | M | Yes | External | Moderate | 4 | No | Yes | No |
| 11 | 3300 | 35+5 | M | No | VH | Severe | 11 | Yes | Yes | Yes |
| 12 | 2100 | 35+5 | M | No | External | Severe | 4 | No | Yes | No |
| 13 | 3300 | 35+5 | M | Yes | External | Severe | 3 | No | Yes | No |
| 14 | 3300 | 41+1 | M | Yes | External | Severe | 11 | Si | Yes | Yes |
| 15 | 4200 | 40+5 | M | Yes | External | Severe | 11 | Yes | No | Yes |
| 16 | 3200 | 41+3 | F | Yes | External | Moderate | 2 | No | Yes | No |
| 17 | 3600 | 38+4 | M | Yes | External | Moderate | 2 | Yes | Yes | No |
| 18 | 3000 | 40 | F | Yes | External | Severe | 8 | Yes | Yes | Yes |
| 19 | 3000 | 41+4 | F | No | External | Moderate | 1 | No | Yes | No |
Degree of HIE based on the semiquantitative scale of Garcia-Alix.
F, female; GA, gestational age; M, male; MRI, magnetic resonance imaging (van Rooij score); VH, Hospital Vall D’ Hebron.
We used the following electrocardiographic definitions, which were based in the European Society of Cardiology guidelines for the interpretation of the neonatal ECG21:
- -
Sinus bradycardia: heart rate less than or equal to 93 beats per minute (bpm) the first day of life, less than or equal to 91bpm up to the third day of life, and less than or equal to 90bpm up to the seventh day of life, inclusive, with a regular heart rhythm;
- -
Normal QTc: value less than 450milliseconds (ms);
- -
Prolonged QTc: calculated value greater than 450ms;
- -
Prolonged PR: intervals lasting more than 140ms.
The presence and severity of significant HIE were classified as moderate or severe using the semiquantitative scale developed by Garcia-Alix.22 All patients underwent a cranial magnetic resonance imaging (MRI) scan between 5 and 7 days of life, with severity graded according to the criteria of van Rooij et al.23
We collected data on variables perceived as potential confounding factors due to their association with QT prolongation in the literature, including serum levels of potassium, magnesium and calcium, and the use of drugs that prolong the QTc interval as well as vasoactive drugs (adrenaline, dopamine and dobutamine).
The study protocol was reviewed and approved by the Clinical Research Ethics Committee of the HUVH, and we safeguarded the confidentiality of all the data obtained from the patients in the study.
Statistical analysisWe conducted a descriptive analysis of demographic and clinical variables and of the results of diagnostic tests. We present categorical variables as percentages and frequencies. Continuous variables (gestational age and weight) are expressed as median and interquartile range.
We compared values of the primary outcome variable, the corrected QTc measured in ms, in relation to subgroups by means of generalised linear models for an accurate analysis of repeated measurements in each participant. Statistical significance was defined as P<.05. We performed the analysis with the statistical software SPSS version 20.0 (IBM Corp, Armonk, USA).
ResultsA total of 19 patients were included in the study, of whom 73.5% (n=14) had transferred from other facilities (with passive hypothermia initiated during transport). Forty-seven percent were male newborns, the median gestational age was 40+2 weeks (36+6–41+1 weeks), and the median weight 3020g (2400–3400g). Ten patients (53%) had moderate HIE based on the clinical assessment (Table 1).
Sentinel events were identified in 53% of patients (n=10), the most frequent of which were placental abruption (n=3) and obstructed labour (n=3). Other documented events were umbilical cord prolapse, uterine rupture, chorioamnionitis or foetal-maternal haemorrhage.
Based on the cranial MRI scan performed between days 5 and 7 of life, eleven patients (58%) had a van Roij score equal to or greater than 4.
All patients were monitored by amplitude-integrated electroencephalography (EEGa) during hypothermia, the rewarming period and the days that followed. Fifty-eight percent of patients (n=11) had clinical/electrical seizures, and all of them received anticonvulsant therapy. The baseline EEGa showed a burst suppression pattern in 32% of patients (n=6), a low voltage pattern in 21% (n=4) and an isoelectric pattern in 21% (n=4).
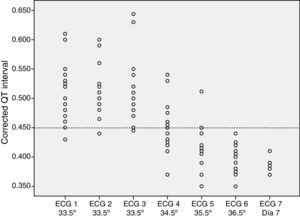
The ECG tracings revealed a prolonged QTc interval in all patients during hypothermia (Fig. 1). In 84% of patients (n=16), this prolongation was detected in all the ECGs performed during hypothermia. In three patients, one of the recordings did not show a prolonged QTc. After rewarming, the QTc interval normalised in all patients.
The analysis of the ECG recordings taken during rewarming showed that at 34.5°C, 47% of the patients (n=9) still had a prolonged QTc interval, and at 35.5°C, 10% of patients (n=2) still had a prolonged QTc interval. None of the patients had a prolonged QTc interval at temperatures above 36°C.
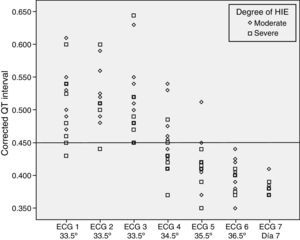
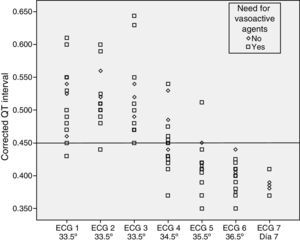
We did not find statistically significant differences in the QTc interval prolongation based on the degree of HIE (P=.192) or the use or lack of inotropic support (P=.669) (Figs. 2 and 3).
None of the patients developed potentially severe arrhythmias. The measured serum levels of potassium, magnesium and calcium were within normal ranges in all patients. There was no documented use of other drugs that could prolong the QT interval (with the exception of the use of vasoactive drugs described above).
Sinus bradycardia was detected at some point during therapeutic hypothermia in 73.5% of patients (n=14) by continuous cardiorespiratory monitoring. Prolongation of the PR interval was detected in none.
One of the patients with moderate HIE developed an isolated episode of paroxysmal supraventricular tachycardia during admission (at 10 days post birth) that required antiarrhythmic therapy, which was discontinued prior to discharge from the unit. We did not find an association between this episode of arrhythmia and the therapeutic hypothermia, nor observed a prolongation of the QTc interval at any point of his stay in the unit after it had normalised with rewarming.
DiscussionThere is a dearth of published evidence on the association of the electrical activity of the heart and therapeutic hypothermia for significant HIE in the neonatal population. The efficacy and safety of this treatment have been demonstrated, and there is cumulative evidence on the development of sinus bradycardia as a frequent and reversible side effect of hypothermia. The association of hypothermia with low blood pressure secondary to the decreased heart rate and requiring inotropic support has also been described. Very few publications address other potential effects of hypothermia in neonates, and they consist of isolated cases that suggest that it may lead to abnormal prolongation of the QTc interval that resolves with rewarming.24,25
This is the first prospective study performed in newborns with significant HIE treated with moderate hypothermia with the purpose of analysing the association of the QTc interval with body temperature in this population.
In all our patients, moderate hypothermia induced a prolonged QTc interval during treatment. Also, the QTc interval normalised in all patients once their body temperatures reached normal physiological ranges (equal to or higher than 36°C).
In any case, the prolongation of the QTc interval in newborns with significant HIE subjected to hypothermia did not seem to increase the risk of developing potentially severe arrhythmias.
It does not seem that potentially confounding factors, such as the use of inotropic support or the degree of the hypoxic-ischaemic insult (severity of encephalopathy), influence the behaviour of the QTc interval in relation to temperature.
Sinus bradycardia is a frequent side effect of hypothermia in newborns, as has been documented in multiple studies and observations. Although studies conducted in adults found a nearly universal prolongation of the PR interval in association with hypothermia, PR intervals greater than 140ms have not been observed in studied patients (none exceeded 120ms).
It could be useful to perform an ECG after rewarming to confirm the presence of a normal QTc interval to avoid missing potential congenital QT interval abnormalities, and this test could be carried out immediately after completing rewarming or at a later time. This ECG can be particularly relevant in survivors of postnatal collapse treated with off-protocol hypothermia, in whom assessment for a pathological arrhythmia as a potential cause of postnatal collapse could be contemplated. We consider that once ECG features are confirmed to be normal after rewarming, it is not necessary to follow up these patients in the cardiology department.
There are some limitations to this study. Due to the lack of studies analysing QTc prolongation in newborns with significant HIE treated with hypothermia in the existing literature, we had to undertake this study as exploratory research with the purpose of increasing our knowledge of this association in newborns and obtaining the necessary data to design future confirmatory studies that would test specific hypotheses and carry out a formal sample size calculation. Since hypothermia must be induced as early as possible in these patients, we could not use each case as its own control, and it was not possible to obtain ECGs prior to cooling.
ConclusionsModerate hypothermia for the treatment of newborns with asphyxia and significant HIE seems to induce a transient prolongation of the QTc interval that resolves with rewarming to normal body temperature (greater than 36°C). At the temperature used in therapeutic (moderate) hypothermia, there seems to be no risk of developing severe ventricular arrhythmias as long as factors such as serum electrolyte levels or the use of drugs that prolong the QTc interval are within normal ranges.
We believe that these patients do not require a followup programme if their QTc interval have normalised after reaching a normal body temperature.
Further and more powerful studies are needed to assess more accurately the association between body temperature and the QTc interval and other electrocardiographic parameters.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Vega L, Boix H, Albert D, Delgado I, Castillo F. El intervalo QT corregido durante la hipotermia terapéutica en la encefalopatía hipóxico-isquémica. An Pediatr (Barc). 2016;85:312–317.