Birth by elective caesarean section in late preterm and early term newborn increases the risk of respiratory distress. Administration of antenatal corticosteroids in these cases could reduce the respiratory distress and its severity.
ObjectivesTo determine the influence of antenatal corticosteroids use in elective caesarean sections in the respiratory distress of the newborn from 35+0 to 38+6 weeks of gestational age.
Patients and methodsRetrospective analytical study of caesarean sections from 35+0 to 38+6 gestational age was conducted in a tertiary hospital from January 2013 to April 2017. Data were collected from medical records of pregnant women and newborns after an implementation of new protocol of betamethasone administration to these elective caesarean sections. Analysis was performed on 2 newborn subgroups: preterm newborn (PTN) 35–36 gestational age and term (TN) 37–38 weeks.
ResultsA total of 208 elective caesarean sections were performed in the study period. Corticosteroids were administered in 97 (46.6%) of cases. The percentage of respiratory distress was higher in the group of preterm newborn compared to term newborn (29% vs. 8.8%, P<.001) and in term newborn higher at a lower gestational age. Between treated with corticosteroids or not, no significant differences were found in the treated and non-treated in the development of respiratory distress (PTN 30 vs. 30%, TN 9.1 vs. 6.9%, P=.6).
ConclusionsNo statistically significant differences were found in this study in favour of the administration of an antenatal dose of betamethasone in the reduction of respiratory distress in the elective caesarean sections from 35+0 to 38+6 gestational age. The delay in the indication of elective caesarean sections, whenever possible, could help reduce the incidence of newborn respiratory distress.
El nacimiento por cesárea programada de los recién nacidos pretérmino tardíos y a término precoces aumenta el riesgo de distrés respiratorio. La administración de corticoides antenatales en estos casos podría disminuir el distrés respiratorio y su gravedad.
ObjetivosDeterminar la repercusión del uso de corticoides antenatales en las cesáreas programadas en el distrés respiratorio del recién nacido de 35 a 38+6 semanas de edad gestacional (SEG).
Pacientes y métodosEstudio analítico retrospectivo en un hospital de tercer nivel, de las cesáreas de 35+0 a 38+6 SEG desde enero de 2013 hasta abril de 2017. Recogida de datos de las historias clínicas de las gestantes y recién nacidos tras la instauración de un nuevo protocolo de administración de una dosis de betametasona a las gestantes con cesáreas programadas de esta edad gestacional. Análisis en 2 subgrupos: recién nacidos pretérmino (RNPT) 35-36 SEG y a término (RNT) 37-38 SEG.
ResultadosEn este periodo se realizaron 208 cesáreas programadas. Se administraron corticoides en 97 casos (46,6%). El porcentaje de distrés fue mayor en el grupo de cesáreas programadas de RNPT comparado con los RNT (29 vs. 8,8%; p<0,001) y entre los RNT fue mayor a menor edad gestacional. Entre las cesáreas programadas tratadas y no tratadas con corticoides no se encontraron diferencias significativas en cuanto al desarrollo de distrés respiratorio (RNPT 30 vs. 30%; p=1; RNT 9,1 vs. 6.9%; p=0,6).
ConclusionesEn este estudio no se encontraron diferencias estadísticamente significativas a favor del beneficio de la administración de una dosis antenatal de betametasona en la disminución del distrés respiratorio del recién nacido en las cesáreas programadas de 35+0 a 38+6 SEG. El retraso en la indicación de cesáreas programadas, siempre que sea posible, podría contribuir a disminuir la incidencia de distrés respiratorio.
Respiratory distress syndrome (RDS) is one of the main causes of early neonatal morbidity and mortality, especially in extremely preterm newborns. Respiratory failure in these patients results from a deficit of surfactant and the immaturity of the lungs and other organs.1–3
The antenatal administration of corticosteroids to pregnant women at risk of preterm delivery between 24 and 34+6 weeks of gestation is currently recommended to accelerate lung maturation and reduce the incidence of RDS, among other beneficial effects.2
Infants born preterm between 35 and 36+6 weeks’ gestation have a higher incidence of respiratory complications compared to infants born preterm due to their immaturity, with a reduced catecholamine response and an increase in lung fluid retention. On the other hand, caesarean delivery is another risk factor for the development of respiratory distress in these preterm infants as well as in term infants delivered before 39 weeks’ gestation, especially in case of planned caesarean delivery in the absence of labour, as exposure to uterine contractions stimulates the clearance of foetal lung fluid.4
Some studies have shown that prophylactic corticosteroid administration before planned caesarean deliveries may reduce the incidence and severity of respiratory distress in late preterm and early full-term infants.3,5
In the current literature, the use of antenatal corticosteroid therapy in newborns delivered preterm after 35 weeks’ gestation remains controversial. The American College of Obstetricians and Gynecologists recommends its use in pregnant women at risk of late preterm birth, independently of the mode of delivery, and does not recommend it in case of caesarean delivery at term.
On the other hand, the Royal College of Obstetricians and Gynaecologists of the United Kingdom does recommend the use of antenatal corticosteroids in planned caesarean deliveries to be performed before 38+6 weeks’ gestation to reduce the risk of neonatal respiratory distress.6
The obstetricians in the hospital that participated in the study presented here, adhering to the latter recommendation, implemented a new protocol in 2013 by which antenatal corticosteroids were administered before planned caesarean deliveries with absence of labour performed between 35+0 and 38+6 weeks’ gestation. The dosage consisted of 1 dose of intramuscular betamethasone given 24h before the caesarean section.
Patients and methodsWe conducted a retrospective analytical study of all planned caesarean deliveries performed between 35+0 and 38+6 weeks’ gestation in a tertiary care hospital from January 2013 to April 2017.
Primary objectiveTo determine the impact of the use of antenatal corticosteroid therapy in cases of planned caesarean delivery between 35+0 and 38+6 weeks’ gestation in the development neonatal respiratory distress.
Secondary objectivesTo analyse the impact of the use of antenatal corticosteroid therapy in the 1- and 5-min Apgar scores, need of conventional mechanical ventilation, need of non-invasive ventilation (NIV), admission to the neonatal intensive care unit (NICU), length of stay, maximum required fraction of inspired oxygen (FiO2) and the development of hypoglycaemia.
We obtained the data by the manual review of the health records of pregnant women, collecting information on the following variables: planned caesarean without labour, administration of corticosteroids, gestational age, chorioamnionitis, amniotic fluid characteristics, maternal diabetes and oligohydramnios. We also reviewed the records of the newborns and collected the following data: sex, Apgar score at 1 and 5min, birth weight, small for gestational age (SGA), admission to ward, admission to NICU, development of respiratory distress, need for mechanical ventilation or NIV, duration of ventilation in hours, maximum FiO2, length of stay in days and hypoglycaemia. We analysed outcomes in 2 subgroups of patients classified according to gestational age at birth: preterm (PT) newborns, delivered at 35 to 36 weeks’ gestation, and full-term (FT) newborns, delivered at 37–38 weeks’ gestation.
We excluded from the study newborns with congenital malformations or transferred to a different hospital.
Definitions- -
Respiratory distress. Presence of clinical signs of respiratory distress due to any of the following causes: RDS, transient tachypnoea, bronchopneumonia.
- -
Hyaline membrane disease or RDS. Presence of clinical signs of respiratory distress requiring supplemental oxygen and evidence of reticulonodular infiltrates in chest radiograph.
- -
Transient tachypnoea: respiratory distress in absence of infiltrates on chest radiograph that resolves spontaneously before 72h post birth.
We analysed the data with the statistical software SPSS version 20.0 for Windows. We defined statistical significance as a P-value of less than .05.
We have summarised quantitative data as mean and standard deviation and qualitative data as frequency distributions.
We analysed the association of qualitative variables by means of the chi square test; if the number of cells with expected values of less than 5 exceeded 20% of the total, we used the Fisher exact test or, for variables with more than 2 categories, the likelihood ratio.
We assessed the normality of the data with the Kolmogorov–Smirnov test. We compared qualitative data by means of the Student t test for independent samples or the Mann–Whitney U test as applicable.
ResultsDuring the study period, there were a total of 285 caesarean deliveries, of which 208 (73%) were planned and performed in the absence of labour.
Of all planned caesarean deliveries, 80.8% were performed at term (37–38 weeks) and 19.2% preterm (35–36 weeks).
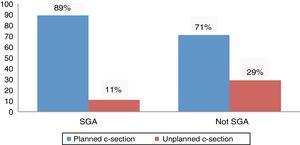
The proportion of planned caesarean delivery was higher in SGA newborns, a difference that was statistically significant (34/38 [89%] vs. 174/245 [71%]; P=.016) (Fig. 1).
Antenatal corticosteroids were given in 97 (46.6%) of the cases of planned caesarean delivery in which they were indicated.
The administration of antenatal corticosteroids where indicated was more frequent in PT newborns compared to FT newborns, a difference that was statistically significant (30/40 [75%] of PT vs. 67/168 [39.9%] of FT newborns; P<.001).
In all gestational age groups, most identified cases of immediate respiratory distress corresponded to transient tachypnoea (20), while the rest corresponded to hyaline membrane disease (2), pneumonia (1), pulmonary hypertension (1) and apparent life-threatening event (1).
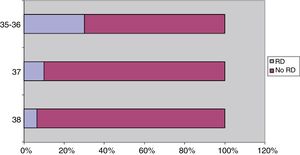
Without taking into account the administration of corticosteroids, the proportion of respiratory distress in cases of planned caesarean delivery was greater in the group of PT infants delivered between 35 and 36 weeks’ gestation compared to FT infants delivered between 37 and 38 weeks, a difference that was statistically significant (respiratory distress, 12/40 PT newborns [30%] vs. 13/168 FT newborns [7.8%]; P<.001), and in the subset born at term, the proportion of respiratory distress was higher in the newborns delivered at 37 weeks compared to those delivered at 38 weeks (respiratory distress, 6/60 of those born at 36 weeks [10%] vs. 7/108 of those born at 38 weeks [6.5%]) (Fig. 2).
The distribution of cases of planned caesarean delivery based on the administration or lack of administration of antenatal corticosteroids was similar when compared based on sex, presence of oligohydramnios, presence of meconium in amniotic fluid and presence of chorioamnionitis in both gestational age groups. There was a higher frequency of antenatal corticosteroid prophylaxis in the group where the planned caesarean delivery was performed at 35 or 36 weeks’ gestation in mothers without diabetes compared to mothers with diabetes (27/33 [81.8%] vs. 3/7 [42.8%]), although this difference was not statistically significant (Table 1).
Characteristics of newborns delivered by planned caesarean section exposed and not exposed to antenatal corticosteroids by gestational age group.
| Gestational age | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 35–36 | 37–38 | ||||||||||
| Corticosteroids | Corticosteroids | ||||||||||
| No | Yes | No | Yes | ||||||||
| n | % | n | % | P | n | % | n | % | P | ||
| Sex of child | 0 | 4 | 40.0 | 17 | 56.7 | .47 | 46 | 45.5 | 31 | 46.3 | .93 |
| 1 | 6 | 60.0 | 13 | 43.3 | 55 | 54.5 | 36 | 53.7 | |||
| Maternal diabetes | No | 6 | 60.0 | 27 | 90.0 | .05 | 82 | 83.7 | 58 | 86.6 | .61 |
| Yes | 4 | 40.0 | 3 | 10.0 | 16 | 16.3 | 9 | 13.4 | |||
| Oligohydramnios | No | 10 | 100.0 | 28 | 93.3 | 1 | 95 | 97.9 | 66 | 98.5 | 1 |
| Yes | 0 | 0.0 | 2 | 6.7 | 2 | 2.1 | 1 | 1.5 | |||
| Chorioamnionitis | No | 10 | 100.0 | 30 | 100.0 | – | 98 | 100.0 | 66 | 98.5 | .41 |
| Yes | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 1 | 1.5 | |||
| Amniotic fluid | 0 | 10 | 100.0 | 29 | 96.7 | 1 | 100 | 99.0 | 64 | 95.5 | .15 |
| 1 | 0 | 0.0 | 1 | 3.3 | 0 | 0.0 | 2 | 3.0 | |||
| 2 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | |||
| 3 | 0 | 0.0 | 0 | 0.0 | 1 | 1.0 | 1 | 1.5 | |||
Sex of child : 0 (male) 1(female) Amniotic fluid: 0 (clear); 1/2/3 (stained +/++/+++)
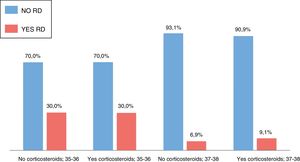
We did not find significant differences in the incidence of respiratory distress between the group treated with antenatal corticosteroids and the group that did not receive this treatment in either of the 2 gestational age groups (Fig. 3). We also did not find significant differences in the need of admission to the ward, admission to the NICU, mechanical ventilation or NIV or in the development of hypoglycaemia. Table 2 presents these results (summary of qualitative variables).
Comparison of qualitative characteristics of newborns delivered by planned caesarean section exposed and not exposed to antenatal corticosteroids by gestational age group.
| Gestational age | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 35–36 weeks | P | 37–38 weeks | P | ||||||||
| Corticosteroids | Corticosteroids | ||||||||||
| No | Yes | No | Yes | ||||||||
| n | n | n | n | ||||||||
| Admission | No | 3 | 30.0 | 9 | 30.0 | 1 | 80 | 79.2 | 49 | 74.2 | .454 |
| Yes | 7 | 70.0 | 21 | 70.0 | 21 | 20.8 | 17 | 25.8 | |||
| RD | No | 7 | 70.0 | 21 | 70.0 | 1 | 94 | 93.1 | 60 | 90.9 | .61 |
| Yes | 3 | 30.0 | 9 | 30.0 | 7 | 6.9 | 6 | 9.1 | |||
| NICU | No | 9 | 90.0 | 24 | 80.0 | .656 | 99 | 98.0 | 65 | 98.5 | 1 |
| Yes | 1 | 10.0 | 6 | 20.0 | 2 | 2.0 | 1 | 1.5 | |||
| MV | No | 9 | 90.0 | 30 | 100.0 | .25 | 101 | 100.0 | 66 | 100.0 | – |
| Yes | 1 | 10.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | |||
| NIV | No | 7 | 70.0 | 22 | 73.3 | 1 | 97 | 96.0 | 60 | 90.9 | .196 |
| Yes | 3 | 30.0 | 8 | 36.7 | 4 | 4.0 | 6 | 9.1 | |||
| Hypoglycaemia | No | 8 | 80.0 | 26 | 86.7 | .629 | 96 | 95.0 | 60 | 90.9 | .229 |
| Yes | 2 | 20.0 | 4 | 13.3 | 5 | 5.0 | 6 | 9.1 | |||
RD,respiratory distress;NICU,neonatal intensive care unit;MV, mechanical ventilation; NIV,non-invasive ventilation
In the comparison of the groups of infants delivered by planned caesarean section exposed and not exposed to antenatal corticosteroids, we did not find any differences in the length of stay, duration of NIV, maximum FiO2 or Apgar score at 1min and 5min in either of the gestational age groups. Table 3 presents these results (summary of quantitative variables).
Comparison of quantitative characteristics of newborns delivered by planned caesarean section exposed and not exposed to antenatal corticosteroids by gestational age group.
| Hours of MV | Maximum FiO2 | Apgar 1 | Apgar 5 | LOS in days | ||||
|---|---|---|---|---|---|---|---|---|
| 35–36GA | Corticosteroids | No | Mean | 17.00 | 22.60 | 8.20 | 9.50 | 6.97 |
| Standard deviation | 39.18 | 3.41 | 1.48 | 0.71 | 9.60 | |||
| Yes | Media | 12.27 | 22.83 | 8.20 | 9.47 | 10.97 | ||
| Standard deviation | 37.46 | 4.49 | 1.42 | 0.68 | 10.10 | |||
| P | .827 | .906 | .889 | .873 | .123 | |||
| 37–38GA | Corticosteroids | No | Media | 1.41 | 21.27 | 8.65 | 9.69 | 1.27 |
| Standard deviation | 12.00 | 1.94 | 0.82 | 0.61 | 3.24 | |||
| Yes | Media | 0.56 | 21.26 | 8.67 | 9.70 | 1.98 | ||
| Standard deviation | 2.88 | 1.30 | 1.20 | 0.60 | 4.35 |
LOS, length of stay; MV, mechanical ventilation; GA, gestational age.
The administration of antenatal corticosteroids in pregnant women at risk of preterm birth before 34+6 weeks’ gestation is currently recommended (grade A1 recommendation) to accelerate lung maturation and reduce the incidence and severity of RDS associated with the deficit of surfactant in these newborns.1,2 Furthermore, antenatal corticosteroids also reduce perinatal and neonatal mortality and the incidence of necrotising enterocolitis and intraventricular haemorrhage. A single course of corticosteroids does not seem to have any short-term adverse effects in either the mother or the newborn.7
Late PT newborns (born at 34–36 weeks’ gestation) had respiratory morbidity more frequently than FT newborns. On the other hand, caesarean delivery is an additional risk factor for the development of respiratory distress both in PT and FT newborns, especially in the absence of labour.3
The usefulness of antenatal steroids in late PR and early FT infants delivered by planned caesarean section is still under debate.
Based on several studies in the literature, antenatal corticosteroid prophylaxis in case of delivery between 34 and 36 weeks’ gestation is associated with a reduction of respiratory morbidity (severe RDS and transient tachypnoea) in the short term, but not with a reduction in mortality.8–10 There are limitations to these studies, some due to their design, others for including infants born at 34 weeks’ gestation in the analysis, others for not differentiating between modes of delivery, not to mention that they use different corticosteroid regimens (selected corticosteroid, number of doses). The data on the long-term effects and outcomes of antenatal corticosteroid administration are scarce.
When it comes to the administration of corticosteroids before performance of planned caesarean sections at term, the most recent Cochrane review found that it can reduce the risk of respiratory distress and admission in to intensive care due to respiratory morbidity, and no difference in relation to the need of conventional mechanical ventilation or mortality. The authors of the review described limitations in the design of these studies and reported that further studies with larger samples would be required to obtain valid results.11
The regimen used in these studies included at least 2 doses of dexamethasone or betamethasone. There are also still few long-term follow-up data on these newborns.12
Thus, there is also no clear evidence on the optimal regimen for antenatal corticosteroid therapy in terms of the type of corticosteroid, number of doses, interval between doses and timing of administration, which have varied between published studies.12–16
The regimen used in the study in our hospital consisted of 1 dose of betamethasone 24h before the planned caesarean delivery at 35+0 to 38+6 weeks’ gestation. Our results differ from those described by other authors.4,8 This may be due in part because other studies have included PT newborns delivered at 34 weeks’ gestation, who are at higher risk of distress compared to newborns with greater gestational ages, and who are more likely to benefit from the prenatal lung maturation induced by corticosteroids. In addition, the regimens used in other studies were different and included 2 doses of betamethasone or dexamethasone, which could have contributed to the differences in the results.
On the other hand, our study has limitations due to its retrospective design and the small sample size in some of the analyses. Another limitation is the lack of universal adherence to the indications for the administration of corticosteroids. This last factor may be due to the initial lack of awareness of the staff after the introduction of the new protocol, as we found that adherence had increased in the last 3 years.
The performance of this study and its findings have contributed, on one hand, to remind staff of the need to adhere strictly to the protocol, and on the other to propose a future prospective study comparing the administration or lack thereof of antenatal corticosteroids or different steroid regimens.
In addition, when we analysed the results of our study we found that, as described in other case series,17 the proportion of patients with respiratory distress and with admission related to planned caesarean section increased with decreasing gestational age in both FT and PT newborns. In our sample, most planned caesarean deliveries occurred in term pregnancies, and delivery by caesarean was more frequent in SGA newborns, probably because SGA was one of the indications for this mode of delivery.
ConclusionsIn our study, we did not find significant differences in the benefit of the administration of a single dose of antenatal corticosteroids in planned caesarean deliveries performed between 35+0 and 38+6 weeks’ gestation for reduction of respiratory distress in newborns.
We also found no differences in the need or duration of NIV, admission to NICU, length of stay, percentage requiring supplemental oxygen, Apgar score at birth or the development of hypoglycaemia.
Further studies are required to determine the most appropriate form and schedule of corticosteroid therapy and the long-term outcomes of its administration in exposed newborns.
In this case series, consistent with previous ones, the development of respiratory distress was significantly more frequent in PT newborns compared to FT newborns, and in the latter subset, more frequent with decreasing gestational age. Therefore, delaying performance of planned caesarean deliveries, where possible, could contribute to decreasing the respiratory distress associated with prematurity and the lack of exposure to labour.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: de la Huerga López A, Sendarrubias Alonso M, Jiménez Jiménez AP, Matías del Pozo V, Álvarez Colomo C, Muñoz Moreno MF. Corticoides antenatales e incidencia de distrés respiratorio del recién nacido en las cesáreas programadas del pretérmino tardío y término precoz. An Pediatr (Barc). 2019;91:415.
Previous presentation: This study was presented as an oral communication and accepted towards the application to become a full member of the Sociedad Española de Neonatología (SENeo) at the Congress of the Sociedad Española de Neonatología held September 27–29, 2017 in Zaragoza, Spain.