Sleep apnoea-hypopnoea syndrome (SAHS) is a disease that affects 2%–4% of the paediatric population.1,2 It consists in the recurrent collapse of the airway during sleep with cessation or reduction of airflow resulting in cycles of hypoxia followed by reoxygenation that in the long term can trigger an inflammatory cascade with systemic effects.2,3
One of the differences between SAHS in children and in adults is the type of repercussions it has on health. Thus, in the paediatric population the most frequent repercussions are growth delay and neurocognitive complications, while daytime somnolence and cardiovascular complications are more frequent in adults.
One of the haematologic changes described in adults with SAHS is an increased haemoglobin concentration,4,5 in some cases in the range of polycythaemia,5 and such changes are believed to be secondary to an increased secretion of erythropoietin resulting from the recurrent hypoxia during sleep. Recently, some authors have started to analyse other factors,5 as not every patient with severe SAHS exhibits these changes.
In addition, there is evidence of a directly proportional correlation between the severity of SAHS and the increase in the red blood cell distribution width (RDW).3 The RDW is currently being investigated as a proinflammatory marker (not only in the context of cardiovascular disease), and probably increases as a result of oxidative stress and chronic inflammation, which cause the release of cytokines that could act on the bone marrow and affect erythropoiesis.3 It has also been hypothesised that this association may be influenced by increased neurohormonal activity.3
Other factors that have been studied in the paediatric population include signs of metabolic disturbances, such as elevation of glycated haemoglobin in patients with SAHS, which is considered a marker of severity (independently of age and weight),6 but the data on RBC laboratory test values are still insufficient. For this reason, we designed a study to assess whether the haematologic changes typically found in adult patients with severe SAHS are also found in children, with the purpose of identifying new screening tools that could help expedite the performance of sleep studies.
We conducted a retrospective study by reviewing clinical health records and preoperatory laboratory test results in patients operated for severe SAHS in our hospital between 2012 and 2016 and in controls of similar age that underwent surgery for other reasons.
We defined severe SAHS as an apnoea-hypopnoea index (AHI) greater than 10 in the sleep study. The variables we analysed were age, AHI or obstructive AHI, oxygen desaturation index (ODI), oxygen saturation nadir, sleep time spent with an oxygen saturation of less than 90% (T90), arousals, total sleep duration, sleep efficiency, percentage of deep sleep and REM sleep and red blood cell (RBC) test values (haemoglobin, haematocrit, mean corpuscular volume and RDW).
We selected a sample of 87 children with severe SAHS (mean age, 4.30 ± 2,27), diagnosed by means of polysomnography in 78 and by means of respiratory polygraphy in 9, and a control group of 88 children (mean age, 6.01 ± 3.68). The mean AHI in the SAHS group was 21.34 ± 12.80. Table 1 summarises the rest of the polygraphy/polysomnography values.
Descriptive analysis of the group of patients with severe SAHS (measures of central tendency and dispersion).
| N | Mean | SD | Median | Q1 | Q3 | |
|---|---|---|---|---|---|---|
| AHI | 87 | 21.34 | 12.80 | 17.70 | 12.90 | 23.90 |
| Nadir | 85 | 79.08 | 10.64 | 83.00 | 74.00 | 87.00 |
| ODI | 87 | 25.45 | 16.09 | 21.70 | 14.65 | 30.75 |
| T90 | 87 | 4.09 | 7.46 | 0.90 | 0.10 | 4.60 |
| Arousals/h | 73 | 13.72 | 9.10 | 11.00 | 7.45 | 18.90 |
| TST, % | 77 | 7.42 | 2.67 | 7.27 | 6.79 | 7.68 |
| N3, % | 70 | 34.81 | 8.59 | 35.10 | 29.30 | 40.62 |
| REMS, % | 70 | 14.27 | 5.19 | 14.75 | 11.00 | 17.85 |
| Efficiency, % | 74 | 86.93 | 7.90 | 88.70 | 83.65 | 92.05 |
AHI, apnoea-hypopnoea index; N3, slow wave sleep; ODI, oxygen desaturation index; REMS, rapid eye movement sleep; SAHS: sleep apnoea-hypopnoea syndrome; SD, standard deviation; T90, time spent with oxygen saturation <90%; TST, total sleep time.
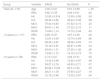
When we compared mean haemoglobin values, we found no significant difference between the SAHS group (12.92 ± 0.92) and the control group (13.09 ± 0.99), nor did we find any differences between groups in any other haematologic variable (Table 2).
Comparison of patients with severe SAHS and controls, overall and stratified by age group.
| Group | Variable | SAHS | No SAHS | P |
|---|---|---|---|---|
| Total (N = 175) | Age | 4.30 ± 2.27 | 6.01 ± 3.68 | < .05 |
| RBC | 4.82 ± 0.35 | 4.80 ± 0.41 | .62 | |
| Hb | 12.92 ± 0.914 | 13.09 ± 0.99 | .24 | |
| Hct | 38.46 ± 2.60 | 39.24 ± 3.09 | .08 | |
| MCV | 79.40 ± 6.83 | 81.52 ± 5.10 | .02 | |
| MCH | 26.87 ± 1.59 | 27.32 ± 2.10 | .11 | |
| RDW | 13.69 ± 1.14 | 13.75 ± 2.44 | .83 | |
| < 6 years (n = 117) | RBC | 4.85 ± 0.37 | 4.67 ± 4.38 | .02 |
| Hb | 12.81 ± 0.94 | 12.74 ± 0.94 | .68 | |
| Hct | 38.26 ± 2.67 | 38.08 ± 2.93 | .74 | |
| MCV | 79.18 ± 3.87 | 80.87 ± 4.96 | .04 | |
| MCH | 26.50 ± 1.51 | 27.05 ± 1.92 | .09 | |
| RDW | 13.92 ± 1.12 | 14.10 ± 3.15 | .70 | |
| ≥ 6 years (n = 58) | RBC | 4.74 ± 0.27 | 4.94 ± 0.31 | .02 |
| Hb | 13.34 ± 0.66 | 13.52 ± 0.87 | .46 | |
| Hct | 39.27 ± 2.18 | 40.63 ± 2.71 | .07 | |
| MCV | 80.26 ± 13.22 | 82.3 ± 5.22 | .53 | |
| MCH | 28.27 ± 1.03 | 27.65 ± 2.27 | .16 | |
| RDW | 12.79 ± 0.68 | 13.32 ± 0.97 | .04 |
Total sample with 175 patients: 87 with severe SAHS (69 aged < 6 years, 18 aged ≥ 6 years) and 88 controls (48 aged < 6 years, 40 aged ≥ 6 years).
Hb, haemoglobin concentration (g/dL); Hct, haematocrit; MCH, mean corpuscular haemoglobin (pg); MCV, mean corpuscular volume (fL); RBC, red blood cell count (×106/μL); RDW, red blood cell distribution width; SAHS: sleep apnoea-hypopnoea syndrome.
Given the significant age difference between groups and the variation in haematologic observed through the different stages of childhood, we decided to carry out an additional analysis by age group (Table 2), which also did not find significant differences in haemoglobin or haematocrit values. In our study, in children aged less than 6 years we found a higher RBC count in the SAHS group compared to the control group, but paradoxically in older children this trend not only did not persist but reversed, with higher RBC counts in the control group, so we were unable to draw conclusions on this aspect. When it came to the RDW, we found that in children aged more than 6 years, RDW values were slightly higher in the control group compared to the SAHS group.
Therefore, in contrast to the adult population, we did not find significant differences in RBC test values in children that underwent surgery for treatment of SAHS compared to children of similar age operated for other reasons.
This is probably due to a lower degree of hypoxia (frequent cases with a T90 of 0% despite significant SAHS) and a shorter duration of the disease. Thus, based on our findings, this is not a useful tool for screening purposes. However, we believe that further research on the subject is required, conducting studies on larger samples, with better matching for age and analysing other comorbidities and clinical parameters (not only haematologic, but also anthropometric and metabolic) that could also have an impact on RBC test values.
Please cite this article as: Rodríguez-Catalán J, López Carnero J, Pérez Tejerizo G, Rodríguez Rodríguez P, Del-Río Camacho G. Análisis de la serie roja en niños con síndrome de apnea-hipopnea del sueño. An Pediatr (Barc). 2020;92:234–236.