Among the environmental factors that can affect the pathological response to gluten in coeliac disease (CD), the factors that influence the immune response, such as infections and use of antibiotics, are proposed. Our objective is to determine the relationship between infections in early life and the risk of CD.
Patients and methodsA retrospective case-control study, including patients aged 0–16 years with a diagnosis of CD was performed between the years 2014–2018. An analysis was made of documented infections in the first 6 months of life, types of infection (respiratory, gastrointestinal, urinary, others), microorganisms involved, and antibiotic therapy used.
ResultsA total of 93 coeliac patients, 93 controls, and 237 infectious episodes were registered. Documented infections affected 67.7% of coeliac patients and 50.5% of controls (P=.017), with a mean of 1.49±1.53 episodes in the coeliac group and 1.05±1.5 in the controls (P=.016). Documented infections in the first 6 months of life doubles the risk of developing CD (OR 2.05; 95% CI; 1.13–3.73), with this risk being higher for respiratory infections, which multiply the risk by 2.3 (OR 2.30, 95% CI; 1.28–4.14). Also, having 3 or more respiratory infections in the first 6 months of life multiplied the risk by 2.8 (OR 2.79, 95% CI; 1.03–7.54). No differences were found related to the types of involved microorganism or regarding the use of antibiotics.
ConclusionsInfections in the first 6 months of life increase the risk of developing CD, especially for respiratory infections and, to a greater extent, if 3 or more episodes occur. The use of antibiotics in this period of life has not been related to an increased risk of CD.
Dentro de los factores ambientales que favorecen la respuesta patológica al gluten en la enfermedad celiaca (EC), se proponen aquellos que modulan el tipo de respuesta inmune, como las infecciones y el uso de antibioterapia. El objetivo de este estudio es determinar la relación entre las infecciones en los primeros meses de vida y el riesgo de EC.
Pacientes y métodosEstudio de casos y controles retrospectivo que incluye pacientes menores de 16 años con diagnóstico de EC entre los años 2014-2018. Se registran las infecciones documentadas en los primeros seis meses de vida clasificadas según el tipo de infección (respiratoria, gastrointestinal, urinaria, otras), microorganismo implicado y antibioterapia utilizada.
ResultadosSe analizaron 93 pacientes celiacos frente a 93 controles con 237 episodios infecciosos que afectaron al 67,7% de los pacientes celiacos y al 50,5% de controles (p=0,017), con una media de 1,49±1,53 episodios en los celiacos y 1,05±1,5 en los controles (p=0,016). La presencia de infección documentada en los primeros seis meses de vida duplica el riesgo de desarrollar EC (OR 2,05; IC95% 1,13-3,73), siendo este riesgo mayor para las infecciones respiratorias, que multiplican por 2,3 el riesgo (OR 2,30; IC95% 1,28-4,14). Además, tener tres o más infecciones respiratorias en los primeros seis meses de vida multiplica por 2,8 el riesgo (OR 2,79; IC95% 1,03-7,54). No se han encontrado diferencias significativas entre ambos grupos respecto al tipo de microorganismo implicado, ni al uso de antibioterapia.
ConclusionesLa presencia de infecciones en los primeros seis meses de vida aumenta el riesgo de desarrollar EC, siendo este riesgo superior para las infecciones respiratorias y, en mayor medida si se producen tres o más episodios. No se ha demostrado que el uso de antibioterapia en dicho periodo se asocie a un aumento del riesgo de EC.
Coeliac disease (CD) is an immune-mediated systemic disorder triggered by the ingestion of gluten in genetically predisposed individuals that manifests with a broad range of signs and symptoms, specific antibodies, a DQ2-DQ8 HLA haplotype associated with susceptibility and characteristic enteropathy.1
The considerable difference between the population with risk haplotypes, which nears 30% in caucasians, and the prevalence of CD, of around 1% in Europe and Spain, entails the necessary involvement of environmental factors in the pathophysiology of the disease.
In recent decades, there has been an increase in the reported prevalence of CD in Western countries associated with an improved understanding of the disease and the application of new diagnostic criteria based on the presence of specific antibodies1,4; but also with the influence of various environmental factors, including the type of delivery, the duration of breastfeeding, socioeconomic status, the pattern of gluten intake, vaccination, history of infection and use of antibiotherapy during childhood, among others.2
When it comes to environmental factors, several studies have found an association between infection in early childhood and the development of CD, although most of these studies are population-based surveys based on questionnaires administered to families5–8 lacking the necessary confirmation of diagnosis by a medical provider or performance of an aetiological or microbiological diagnosis. There is also evidence of the involvement of the microbiota in the development of gastrointestinal and autoimmune diseases, including CD,9,10 and on the changes in the microbiota secondary to antibiotic treatment in the development of CD.11–13
The aim of our study was to assess the association between infections and the use of antibiotherapy in the first 6 months of life and the development of CD.
Patients and methodsWe conducted a retrospective case-control study in a sample of cases of CD diagnosed 2014 and 2018 at the Hospital Universitario Marqués de Valdecilla in Santander, Spain, a tertiary care hospital whose Paediatric Gastroenterology Unit is the referral unit for the entire autonomous community of Cantabria.
We identified cases through the register of patients with coeliac disease of the paediatric gastroenterology unit. We selected patients aged less than 16 years that received a diagnosis of CD in the 2014–2018 period included in one of the caseloads of the unit, chosen at random. We included cases confirmed in adherence to the diagnostic criteria of the European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) at the time,1 and excluded patients with chronic diseases and immunodeficiencies, including isolated IgA deficiency. For each case, we randomly selected a control matched for age and sex by searching the register of patients of the public health system of Cantabria. The criteria used in the search was the date of birth and sex of the cases, and we selected the first match in alphabetical order and applying the exclusion criteria.
We analysed demographic variables, the type of nutrition in the first months of life and enrolment in childcare services. For cases of CD, we collected the age at diagnosis, the type of diagnosis and the class II human leucocyte antigen (HLA) haplotype. Human leucocyte antigen testing was not performed in controls due to the study design and ethical considerations. To identify episodes of infection, we reviewed the electronic health care records of the primary care and speciality care systems. We classified as infection any diagnosis that could be due to infection made in the first 6 months of life by a medical professional (paediatrician or family physician) based on the codes of the International Classification of Diseases, 10th revision in Spanish (CIE-10-ES), in either the primary care system or the emergency departments of hospitals in Cantabria. For each case of infection, we collected data on the type of infection, age at diagnosis, number of infections, involved microorganisms and use of antibiotherapy.
When it came to the type of infection, based on the CIE-10 ES code, we classified cases of acute upper respiratory infection, influenza, pneumonia, acute bronchitis, bronchiolitis and unspecified acute lower respiratory infection as respiratory infections. Similarly, we classified cases of cases of nonsuppurative, suppurative or unspecified otitis media as otitis media, intestinal infectious diseases as gastroenteritis, acute pyelonephritis as urinary tract infection, and lastly other diagnoses coded as other or unspecified infectious disease as febrile syndrome.
The statistical analysis was performed with the software SPSS version 20.0, using the chi square and Fisher exact tests to compare categorical data and the Mann–Whitney U test to compare quantitative data. We also performed a binary logistic regression analysis. In every test, statistical significance was defined as a P-value of less than .05.
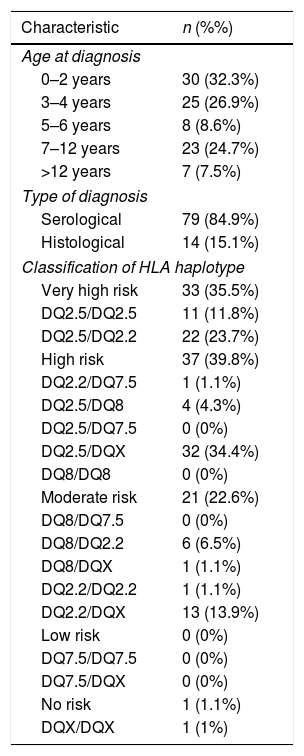
ResultsThe study included 93 paediatric patients given a diagnosis of CD in our unit between 2014 and 2018 that we compared with 93 healthy controls matched for age and sex, resulting in a total sample of 186 individuals with a predominance of the female sex (66.7%). When it came to the cases, the median age at diagnosis of CD was 36 months (interquartile range [IQR], 23–105.5), and most (84.9%) were diagnosed without a biopsy. Table 1 presents the main characteristics of the group of patients with CD, including the risk classification based on the HLA haplotype.3,14
Characteristics of the sample of patients with coeliac disease. The classification of HLA haplotypes was based on the study in the Spanish population conducted by Martínez-Ojinaga.3
| Characteristic | n (%%) |
|---|---|
| Age at diagnosis | |
| 0–2 years | 30 (32.3%) |
| 3–4 years | 25 (26.9%) |
| 5–6 years | 8 (8.6%) |
| 7–12 years | 23 (24.7%) |
| >12 years | 7 (7.5%) |
| Type of diagnosis | |
| Serological | 79 (84.9%) |
| Histological | 14 (15.1%) |
| Classification of HLA haplotype | |
| Very high risk | 33 (35.5%) |
| DQ2.5/DQ2.5 | 11 (11.8%) |
| DQ2.5/DQ2.2 | 22 (23.7%) |
| High risk | 37 (39.8%) |
| DQ2.2/DQ7.5 | 1 (1.1%) |
| DQ2.5/DQ8 | 4 (4.3%) |
| DQ2.5/DQ7.5 | 0 (0%) |
| DQ2.5/DQX | 32 (34.4%) |
| DQ8/DQ8 | 0 (0%) |
| Moderate risk | 21 (22.6%) |
| DQ8/DQ7.5 | 0 (0%) |
| DQ8/DQ2.2 | 6 (6.5%) |
| DQ8/DQX | 1 (1.1%) |
| DQ2.2/DQ2.2 | 1 (1.1%) |
| DQ2.2/DQX | 13 (13.9%) |
| Low risk | 0 (0%) |
| DQ7.5/DQ7.5 | 0 (0%) |
| DQ7.5/DQX | 0 (0%) |
| No risk | 1 (1.1%) |
| DQX/DQX | 1 (1%) |
DQX represents an HLA allele not associated with risk.
The type of nutrition and enrolment in childcare in the CD group and the control group were comparable. In the first 3 months of life, 55.6% of patients with CD and 55.1% of controls received exclusive or predominant breastfeeding, and breastfeeding lasted more than 6 months in 29.6% of patients with CD and 38.5% of controls, with no significant differences (P=1.0 and P=.25, respectively). We also found no significant differences in attendance to childcare services in the first 6 months of life (cases, 6.5%; controls, 5.4%; P=.86).
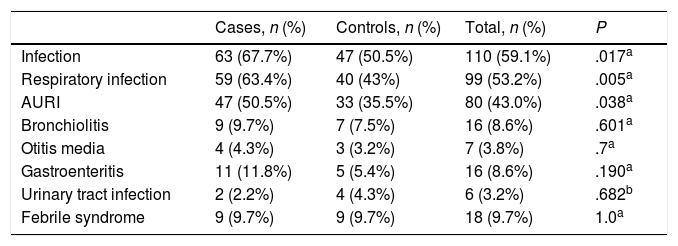
A total of 237 episodes of infection were recorded in the first 6 months of life in 110 of the 186 patients under study (59.1%). Respiratory infections were most frequent (76.8%), followed by fever without febrile syndrome (8.4%), acute gastroenteritis (7.2%), urinary tract infection (2.5%) and other types of infections (5%). In 95.3% of infections, the causative agent was not documented, as the illnesses were mild and self-limited, and antibiotherapy was used in 16%.
In the comparative analysis, we found that infections were more frequent in the CD group, as 67.7% of these patients had documented infections in the first 6 months of life compared to 50.5% of controls (P=.017). The mean number of infections was higher in the CD group compared to the control group (1.49±1.53 vs. 1.05±1.5; P=.016), however, we did not find any differences between groups in terms of the age at the first infection, with a median age of 2 months (IQR, 1–4) in both groups (P=.782). When we compared groups based on the type of infection, we found a higher frequency of respiratory infections in cases compared to controls (63.4% vs. 43%, P=.038), with a higher incidence of upper respiratory tract infections (50.5% vs. 35.5%; P=.038). Acute gastroenteritis was more frequent in the case versus the control group, although the difference was not statistically significant (11.8% vs. 4.3%; P=.19). There were no significant differences in any of the other types of infection (Table 2).
Comparative analysis of infections detected in the first 6 months of life. We report the results for the total cases and the analysis by type of infection. For respiratory infections, we also made specific analysis of acute upper respiratory infection (AURI) and bronchiolitis.
| Cases, n (%) | Controls, n (%) | Total, n (%) | P | |
|---|---|---|---|---|
| Infection | 63 (67.7%) | 47 (50.5%) | 110 (59.1%) | .017a |
| Respiratory infection | 59 (63.4%) | 40 (43%) | 99 (53.2%) | .005a |
| AURI | 47 (50.5%) | 33 (35.5%) | 80 (43.0%) | .038a |
| Bronchiolitis | 9 (9.7%) | 7 (7.5%) | 16 (8.6%) | .601a |
| Otitis media | 4 (4.3%) | 3 (3.2%) | 7 (3.8%) | .7a |
| Gastroenteritis | 11 (11.8%) | 5 (5.4%) | 16 (8.6%) | .190a |
| Urinary tract infection | 2 (2.2%) | 4 (4.3%) | 6 (3.2%) | .682b |
| Febrile syndrome | 9 (9.7%) | 9 (9.7%) | 18 (9.7%) | 1.0a |
The logistic regression analysis showed that a documented history of infection in the first 6 months of life was associated with a 2-fold increase in the risk of developing CD (OR, 2.05; 95% CI, 1.13–3.73), which was greater for respiratory infections, with a 2.3-fold increase in risk (OR 2.30; 95% CI, 1.28–4.14). We also found that having 3 or more respiratory infections in the first 6 months of life was associated with a 2.8-fold increase in the risk of CD (OR 2.79; 95% CI, 1.03–7.54).
The involved pathogens were documented in 6.4% of cases and 5.4% of controls. In the CD group, we found records of infection by adenovirus, enterovirus, Escherichia coli, Moraxella catarrhalis and pneumococcus (2 patients). In the control group, the identified microorganisms were respiratory syncytial virus (RSV), influenza A, Haemophilus influenzae and E. coli (2 patients). We did not find statistically significant differences in the distribution of causative agents between groups (P=.379).
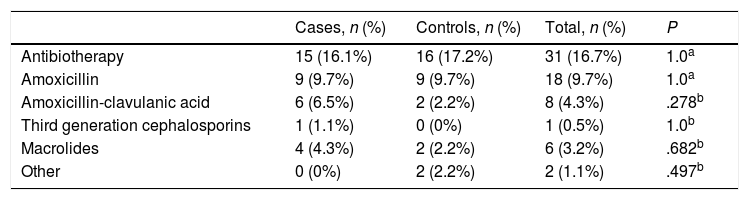
As for the use of antibiotherapy, we did not find significant differences between groups, and at least 1 antibiotic agent had been used in 16.1% of cases and 17.2% of controls (P=1.0). In the entire sample, 7 patients (3.8%) received 2 or more courses of antibiotherapy, corresponding to 5 cases (5.4%) and 2 controls (2.2%), although the difference was not statistically significant. We also found no differences in the type of antibiotic used (Table 3), and Amoxicillin was the agent used most frequently, prescribed to 9.7% of cases and of controls.
Comparison of the use of antibiotherapy in the first 6 months of life. Results for the overall use of antibiotherapy and the antibiotic agents used most frequently.
| Cases, n (%) | Controls, n (%) | Total, n (%) | P | |
|---|---|---|---|---|
| Antibiotherapy | 15 (16.1%) | 16 (17.2%) | 31 (16.7%) | 1.0a |
| Amoxicillin | 9 (9.7%) | 9 (9.7%) | 18 (9.7%) | 1.0a |
| Amoxicillin-clavulanic acid | 6 (6.5%) | 2 (2.2%) | 8 (4.3%) | .278b |
| Third generation cephalosporins | 1 (1.1%) | 0 (0%) | 1 (0.5%) | 1.0b |
| Macrolides | 4 (4.3%) | 2 (2.2%) | 6 (3.2%) | .682b |
| Other | 0 (0%) | 2 (2.2%) | 2 (1.1%) | .497b |
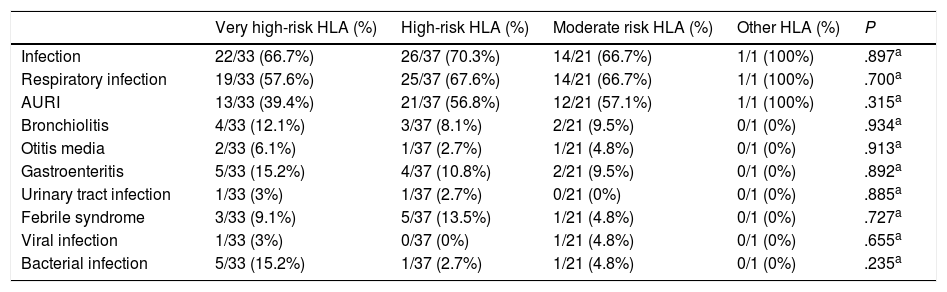
Last of all, in the CD group, we assessed the association of the HLA haplotype with the susceptibility to infection by comparing the incidence of infection in patients with very high-risk, high risk, moderate risk or no-risk HLA haplotypes, and found no significant differences based on this factor (Table 4). We also found no differences when we compared patients homozygous for the DQ2.5 allele to the remaining haplotypes. We were not able to include the haplotypes of the controls in the analysis, as due to the study design we did not have that information.
Comparison of infections detected in the first 6 months of life in the group of patients with a known HLA haplotype (n=92).
| Very high-risk HLA (%) | High-risk HLA (%) | Moderate risk HLA (%) | Other HLA (%) | P | |
|---|---|---|---|---|---|
| Infection | 22/33 (66.7%) | 26/37 (70.3%) | 14/21 (66.7%) | 1/1 (100%) | .897a |
| Respiratory infection | 19/33 (57.6%) | 25/37 (67.6%) | 14/21 (66.7%) | 1/1 (100%) | .700a |
| AURI | 13/33 (39.4%) | 21/37 (56.8%) | 12/21 (57.1%) | 1/1 (100%) | .315a |
| Bronchiolitis | 4/33 (12.1%) | 3/37 (8.1%) | 2/21 (9.5%) | 0/1 (0%) | .934a |
| Otitis media | 2/33 (6.1%) | 1/37 (2.7%) | 1/21 (4.8%) | 0/1 (0%) | .913a |
| Gastroenteritis | 5/33 (15.2%) | 4/37 (10.8%) | 2/21 (9.5%) | 0/1 (0%) | .892a |
| Urinary tract infection | 1/33 (3%) | 1/37 (2.7%) | 0/21 (0%) | 0/1 (0%) | .885a |
| Febrile syndrome | 3/33 (9.1%) | 5/37 (13.5%) | 1/21 (4.8%) | 0/1 (0%) | .727a |
| Viral infection | 1/33 (3%) | 0/37 (0%) | 1/21 (4.8%) | 0/1 (0%) | .655a |
| Bacterial infection | 5/33 (15.2%) | 1/37 (2.7%) | 1/21 (4.8%) | 0/1 (0%) | .235a |
AURI, acute upper respiratory infection.
Our study make a relevant contribution to the current debate on the environmental factors associated to the pathogenesis of CD. Infections detected in the first 6 months of life are a risk factor for developing CD, and this association is stronger in case of respiratory infections. With the data obtained in the study, we could not establish whether gastroenteritis or antibiotherapy are among the factors at play.
Coeliac disease is a disorder with a genetic basis influenced by environmental factors. For decades, and especially after the “Swedish epidemic” of the 1980s, during which the incidence of CD in children aged less than 2 years tripled, there has been evidence of the importance of environmental factors in the pathogenesis of this disorder.15,16 During this epidemic, evidence emerge of a seasonal incidence pattern, with a significantly higher incidence of CD in children born in summer in who gluten was introduced in the winter months.17 Since then, several population-based studies have described an association between infections in early childhood and the development of CD.5–8,11,18
Based on our data, the detection of infection in the first 6 months of life increases the risk of CD by a factor of 2, with an even higher increase in the case of respiratory infections, associated with a 2.3-fold risk, and especially in patients that experience 3 or more episodes, in which case the risk increases by a factor of 2.79. However, we did not find an association between the development of CD and gastrointestinal infections, which may have been due to the low incidence of these infections in the sample.
Infections may contribute to the pathogenesis of CD through various mechanisms. The increased intestinal permeability caused by gastrointestinal infections may facilitate the epithelial translocation of gluten that triggers the inflammatory response.19 In addition, many viral respiratory infections in children manifest with gastrointestinal symptoms,20 suggesting a degree of intestinal involvement that may result in increased permeability. In addition, viruses can play a role through the activation of proinflammatory mechanisms, the production of interferon21 and the activation of tissue enzyme transglutaminase,22 as indicated by the transient elevation of anti-transglutaminase antibodies that may be observed in patients without CD that experience viral infections.23,24
In agreement with our findings, other authors have described an association between different types of infections in early childhood and the subsequent development of CD. In a review of the health records of a sample of 853 patients with CD in Germany, Beyerlein et al.18 found an increased risk in patients that had experienced a gastrointestinal infection (hazard ratio [HR], 1.32) or respiratory infection (HR, 1.22) in the first year of life, and that repeated respiratory or gastrointestinal infections had an increased cumulative risk. Similarly, in their study in 373 Swedish patients with CD, Myléus et al.6 found that a history of 3 or more infections in the first 6 months of life reported by the family increased the risk of CD (OR, 1.5), and the study by Marild et al.7 on 581 Norwegian patients with CD found that a history of more than 10 infections in the first 18 months of life reported by the family is associated with an increased risk of CD compared to experiencing fewer than 4 infections (OR, 1.32), with a stronger association for respiratory infections, as seen in our study. Other authors, such as Kemppainen et al.8 in a study based on questionnaires administered to the relatives of 732 patients with CD in Europe and the United States, have found an increased risk of CD in genetically predisposed children associated with gastrointestinal infections (HR, 1.33), which was consistent with the findings of a study conducted in Italy by Canova et al.11 in 1227 patients with CD in which diagnosed gastrointestinal infections requiring hospital admission were associated with the future diagnosis of CD (incidence rate ratio [IRR], 2.04). In opposition to our own findings and those of the studies we just mentioned, a population-based study conducted in Sweden by Welander et al.5 that identified 44 patients with CD found no significant differences in risk based on the history of gastrointestinal infection or other types of infection after adjusting for the age at the time of gluten introduction and the duration of breastfeeding.
When it came to the specific pathogens involved, we did not find significant differences in our sample, probably because they were identified in few cases. Although the evidence on the subject is scarce, some studies have found an association between infection by rotavirus,25 enterovirus,26 reovirus or norovirus27,28 and the development of CD, and some authors have even proposed that vaccination against rotavirus may have a protective effect, although the evidence is inconsistent.8,29 Simre et al.30 analysed infections by enterovirus, norovirus, parechovirus, rhinovirus, Epstein–Barr virus, cytomegalovirus and adenovirus detected in blood, respiratory secretion and stool samples, and did not find a significant association. The mechanisms by which microorganisms, chiefly viruses, can promote development of a pathological response to gluten are numerous and are not well understood. The proposed mechanisms range from superantigens31,32 to modifications to the innate immune response.28,33,34 As regards the pathogenesis of CD, it has been hypothesised that infections may induce activation of intraepithelial lymphocytes and alter the intestinal permeability, triggering proinflammatory mechanisms and causing changes in the microbiota, which is altered in patients with CD.35–41 In relation to this, other factors that affect the microbiome, such as the HLA haplotype,40 the type of delivery,42,43 the use of proton pump inhibitors44 and the use of antibiotics,12 among others, constitute an important field of research due to their potential involvement in the aetiology and pathogenesis of CD.
Our study did not find evidence of an association between antibiotherapy in the first 6 months of life and the risk of CD, as the use of antibiotics was similar in cases and controls. However, and while the difference was not statistically significant, we found that patients with CD received a greater number of repeated courses of antibiotics compared to healthy controls. This was consistent with the findings of the aforementioned study by Myléus et al.6 in the Swedish population, which did not find an association between the use of antibiotics in the first 6 months of life reported by parents and the development of CD, and of the study by Kemppainen et al.,8 which did not find an association between the use of antibiotherapy reported by parents and the positive serological test for CD in the 4-year follow-up in patients with high-risk HLA alleles in the United States and Europe. On the other hand, the study published in 2019 by Dydensborg et al.12 conducted in a cohort of patients in Sweden and Denmark (3346 cases) suggests that the use of systemic antibiotherapy in the first year of life is associated with CD (OR, 1.26) with a stronger association the greater the number of antibiotics prescribed, with no differences based on the type of antibiotic. Previously, in a cohort of 2933 Swedish children with CD, Marild et al.13 had reported that the use of systemic antibiotics was associated with the subsequent development of CD (OR, 1.40), and Canova et al.11 found similar results in a cohort of Italian children in which the use of antibiotics in the first 12 months of life reported by parents was associated with CD (IRR, 1.24), with the incidence increasing with the number of prescribed courses of antibiotics and with the use of cephalosporins. As Caminero and Verdu suggest,35 it is possible that differences in the type of antibiotic, dose and frequency of administration influence the results, making them difficult to interpret.
As for the involvement of the HLA haplotype in the susceptibility to infection, since class II HLAs participate in antigen presentation, it is possible that the increased incidence of infection in the group of patients with CD could be due to specific HLA alleles. To resolve this concern, we compared the HLA-II haplotypes of patients with CD and concluded that there were no differences in the incidence of infection based on haplotype. For ethical reasons, we did not perform HLA testing in controls, which diminished the power of the analysis.
This study has the limitations intrinsic in retrospective designs (limited availability of information and interrater variability in the criteria used for diagnosis of infection) and its sample size, in addition to the potential for reverse causality, that is, for CD causing symptoms suggestive of infection or increasing the risk of infection; however, we minimised the latter risk by restricting the analysis to the first 6 months of life, before the development of CD. Among the strengths of the study, we would like to highlight that this is the first study of this nature carried out in the Spanish population, based not on parental reports but on diagnoses of infection made by health professionals, with performance of microbiological tests when applicable, and that analysed individual antibiotic prescriptions.
Despite the aforementioned limitations, we may conclude that infections in the first 6 months of life do increase the risk of developing CD, with a higher risk associated with respiratory infections and with experiencing 3 or more episodes of infectious illness. We did not find evidence supporting an association of antibiotherapy with an increase in the risk of CD. To improve our knowledge of the aetiology of this disease, further research is necessary, with performance of prospective multicentre studies in larger samples analysing the association between different types of infections and specific causative agents and antibiotics, as well as the association with other factors, including the genetic makeup, the microbiota and the diet.
FundingThe study did not receive any form of funding.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Llorente Pelayo S, Palacios Sánchez M, Docio Pérez P, Gutiérrez Buendía D, Peña Sainz-Pardo E, Vega Santa-Cruz B, et al. Infecciones en la primera infancia como factor de riesgo de enfermedad celiaca. An Pediatr (Barc). 2021;94:293–300.
Previous presentation: this study was presented at the XXVI National Congress of the Sociedad Española de Gastroenterología, Hepatología y Nutrición Pediátrica; May 16–18, 2019; Santander, Spain.