Survival after hematopoietic stem cell transplantation has improved dramatically in recent years. Unfortunately, there is an increased risk of subsequent malignant neoplasms (SMN) in this population and this represents a significant cause of late mortality.
Patients and methodsIn this study, we analysed the incidence of SMN and the associated risk factors in patients referred at a paediatric age for hematopoietic stem cell transplantation (allogeneic or autologous) in our centre.
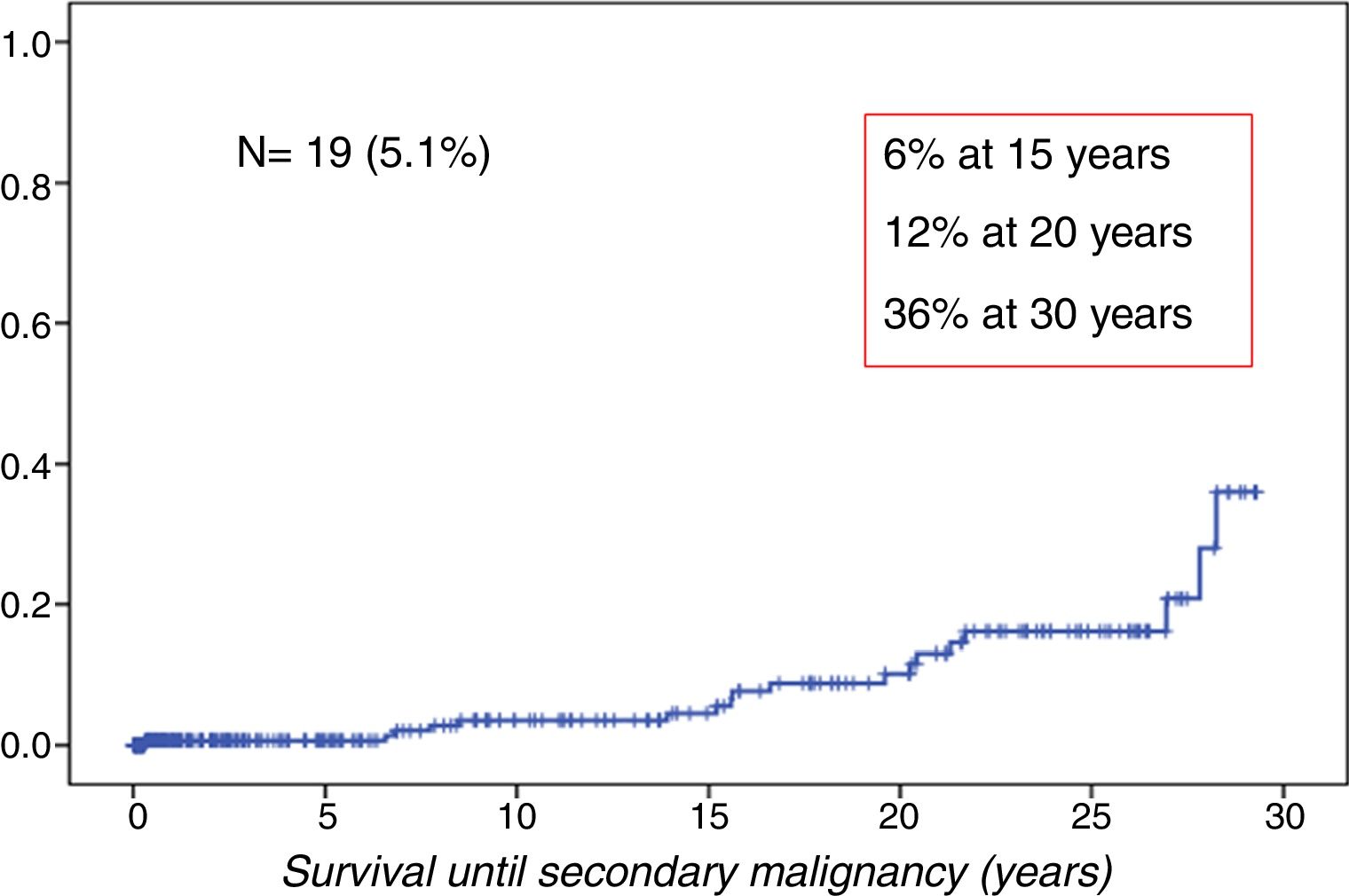
ResultsWe observed 19 cases of SMN in a cohort of 371 patients, with a cumulative incidence of 6, 12, and 36% at 15, 20, and 30 years of follow-up, respectively. The solid tumours were the most prevalent malignancies. The risk was significantly higher than expected in the general population for each tumour type and in the different age ranges (P<.0001). Radiotherapy and chronic GvHD were the main risk factors for the development of SMN in our series.
ConclusionsWe observed a high incidence of SMN among hematopoietic stem cell transplantation survivors highlighting the need for life-long surveillance.
La supervivencia después del trasplante de progenitores hematopoyéticos ha mejorado de forma importante en los últimos años. Desafortunadamente, se ha descrito un mayor riesgo de neoplasias malignas secundarias (NMS) en esta población representando una causa importante de mortalidad tardía.
Pacientes y métodosEn este estudio, analizamos la incidencia de NMS y los factores de riesgo asociados en los pacientes sometidos en edad pediátrica a un trasplante de progenitores hematopoyéticos (alogénico o autólogo) en nuestro centro.
ResultadosObservamos 19 casos de NMS en una cohorte de 371 pacientes, siendo la incidencia acumulada del 6, 12 y 36% a los 15, 20 y 30 años de seguimiento, respectivamente. Los tumores sólidos fueron los más prevalentes. El riesgo fue significativamente mayor de lo esperado en la población general para cada tipo de tumor y en los diferentes rangos de edad (p<0,0001). La radioterapia y la EICH crónica fueron los principales factores de riesgo para el desarrollo de NMS en nuestra serie.
ConclusionesHemos observado una alta incidencia de neoplasias malignas secundarias en los supervivientes de un trasplante de progenitores hematopoyéticos evidenciando la necesidad de un seguimiento a largo plazo de esta población de pacientes.
Hematopoietic stem cell transplantation (HSCT) can cure several malignant and nonmalignant diseases, congenital and acquired, in paediatric and adult patients. In recent years, advances in the procedure and in associated supportive therapies have resulted in an increase in long-term survival. Unfortunately, an increased risk of subsequent malignant neoplasms (SMN) has also been described in this group of patients. There are few reports in which the studied population is exclusively paediatric. The studies of Curtis et al.1 in 1997 and the study of Socié et al.2 in 2000 reported that the risk of SMN was 36-fold and 45-fold, respectively, in children that had undergone HSCT. Both studies found an inverse correlation between the risk of a late malignancy and age.
In our study, we analysed the incidence of SMN in patients that underwent HSCT in our paediatric unit and the associated risk factors. Analysing the risk factors related to the development of these malignancies is crucial for the purpose of improving the conditioning regimens and supportive therapies associated with HSCT.
Patients and methodsThe cohort under study comprised 371 patients that underwent HSCT (autologous or allogeneic) between 1984 and 2013 at the Paediatric HSCT Unit of our hospital. We analysed a total of 434 transplants, which amounted to all the HSCTs performed in our unit between 1984 and 2013. There were no losses to follow-up. Of the 371 patients, 63 received more than one transplant: 2 HSCTs (n=58) or 3 HSCTs (n=5). We obtained the informed consent of the legal guardians for the HSCT procedure and subsequent studies. We collected data on patient characteristics, HSCT type, and clinical outcomes prospectively in a database designed for the purpose. We finished collecting outcome data on September 30, 2016. The duration of follow-up was 30 years. We contacted all adult patients discharged from follow-up at our hospital to obtain information about their current vital status and complications such as malignancies. All the secondary malignancies had been diagnosed in our hospital, so we were able to review the pertinent diagnostic test and patient health records. The median age of the patients at the time of transplantation was 8.4 years (0.1–20 years), and 9 patients were aged more than 18 years. Of all patients, 226 (60.9%) were male and 145 (39.1%) female.
The indication for HSCT was a malignant disease in 317 patients (85.4%) and a nonmalignant disease in 54 patients (14.6%). The malignant diseases included blood tumours (n=209) and solid tumours (n=108). The nonmalignant diseases included primary immunodeficiencies (n=20), aplastic anaemias (n=11), congenital bone marrow failures (n=8), hemoglobinopathies (n=8) and metabolic diseases (n=7). The disease status at the time of transplantations in patients with malignant disease was first complete remission in 146 patients, second complete remission in 101 patients and advanced stage or refractory disease in 7 patients. The HSCT was autologous in 234 cases (53.9%) and allogeneic in 200 cases (46.1%). The donor was a related donor in 141 cases (32.5%) and an unrelated donor in 59 cases (13.6%). As for HLA compatibility, the donor of allogeneic transplants was a matched sibling donor in 106 cases, a haploidentical donor in 35 cases, a matched unrelated donor in 23 cases, and a mismatched unrelated donor in 36 cases.
The most frequent source of hematopoietic stem cells was bone marrow (68.7%; n=298) followed by mobilized peripheral blood (23.7%; n=103) and umbilical cord blood (5.8%; n=25). A combination of different sources (cord blood plus bone marrow or peripheral blood) was used in 8 transplants. The conditioning regimen was based on total body irradiation (TBI) in 48.2% of the HSCTs (n=209). The fractionated irradiation dose ranged between 10 and 13.5Gy in most cases (n=190). Few patients received a dose lower than 10Gy (n=12) or higher than 13.5Gy (n=7). The conditioning regimens based on chemotherapy included busulfan and cyclophosphamide±other chemotherapeutic (CTx) agents (n=84), cyclophosphamide+other CTx agents (n=62), fludarabine+other CTx agents (n=46) and etoposide+other CTx agents (n=26).
Seven patients did not receive any conditioning treatment. Fifty-eight patients had received radiotherapy (RT) before HSCT as treatment for their malignant disease. The RT dose generally ranged from 12.5Gy to 20Gy (n=47) and exceeded 20Gy in 11 patients.
Graft-versus-host disease (GvHD) prophylaxis was based mainly on cyclosporine, alone (n=88) or in association with other immunosuppressant drugs, such as methotrexate±mycophenolate (n=70), prednisone (n=18) or mycophenolate (n=18). In vivo T-cell depletion was performed in 56 HSCTs by means of antithymocyte thymoglobulin (ATG) or alemtuzumab (Campath).
DefinitionsWe defined event as any adverse occurrence after the transplant: disease relapse, secondary malignancy, or death.
We defined overall survival as survival from the time of transplantation, and event-free survival as duration of survival from the time of transplantation with no evidence of any event. We defined transplant-related mortality as death from any cause related to transplantation with the exception of the underlying disease.
Statistical analysisWe used the log-rank test to analyse potential risk factors. We performed the univariate analysis by constructing contingency tables and applying the inferential chi square test. We performed the multivariate analysis by developing a logistic regression model, calculating the odds ratios for each factor. The statistical significance level was fixed at the usual 5% (α=0.05). All the analyses were performed with the statistical package IBM-SPSS (Statistical Package for Social Sciences) version 22.0 for Windows.
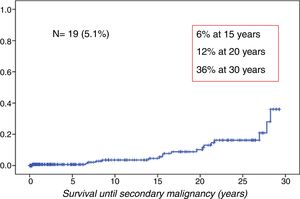
ResultsWe observed 19 cases of SMN in the cohort (5.1%). The cumulative incidence was 6% at 15 years, 12% at 20 years and 36% at 30 years of follow-up (Fig. 1).
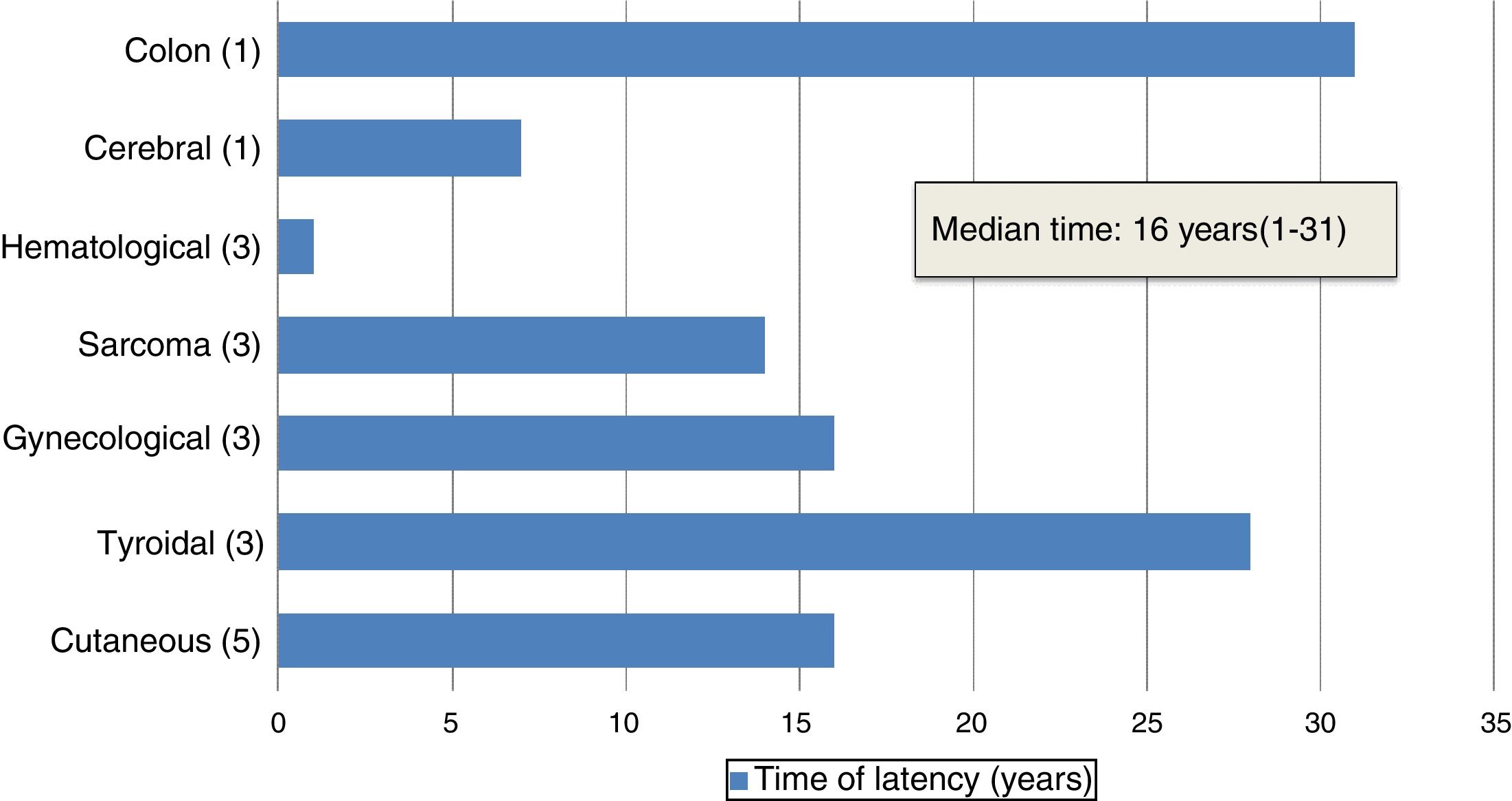
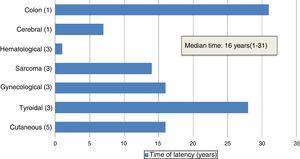
The median age of patients at diagnosis of the SMN was 25 years (11–42 years). The median duration of the latency period between HSCT and diagnosis of the SMN was 16 years (1–31 years) (Fig. 2).
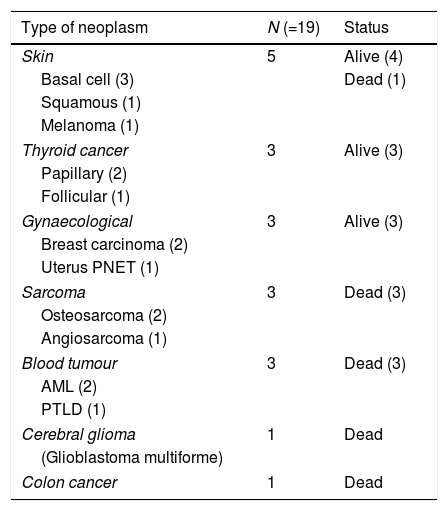
In our series, solid tumours were the most prevalent malignancies, amounting to 84% of cases (n=16). The median latency period for solid tumours was 18.5 years (7–28 years). The secondary solid tumour was the cause of death in 5 patients. Skin cancers were the most frequent type of solid tumour (n=5), followed by carcinomas of the thyroid (n=3), gynaecological cancers (n=3) and sarcomas (n=3). There was one case of brain tumour and one case of colon cancer. Blood tumours amounted to 16% of the cases in our series (n=3): 2 cases of acute myeloblastic leukaemia (AML) and 1 case of post-transplant lymphoproliferative disorder (PTLD). All three patients died of the secondary malignancy. The latency period was shorter compared to solid tumours: less than 1 year for 2 of the patients (Table 1).
Types of subsequent malignant neoplasms.
| Type of neoplasm | N (=19) | Status |
|---|---|---|
| Skin | 5 | Alive (4) |
| Basal cell (3) | Dead (1) | |
| Squamous (1) | ||
| Melanoma (1) | ||
| Thyroid cancer | 3 | Alive (3) |
| Papillary (2) | ||
| Follicular (1) | ||
| Gynaecological | 3 | Alive (3) |
| Breast carcinoma (2) | ||
| Uterus PNET (1) | ||
| Sarcoma | 3 | Dead (3) |
| Osteosarcoma (2) | ||
| Angiosarcoma (1) | ||
| Blood tumour | 3 | Dead (3) |
| AML (2) | ||
| PTLD (1) | ||
| Cerebral glioma | 1 | Dead |
| (Glioblastoma multiforme) | ||
| Colon cancer | 1 | Dead |
The indication for transplantation in almost every patient that developed a SMN (17 out of 19 patients) was a malignancy: acute leukaemia in 12 patients and a solid tumour in 5 patients. The non-malignant disorders that indicated HSCT were Fanconi anaemia and aplastic anaemia.
Eleven patients underwent autologous transplantation and 8 patients allogeneic transplantation.
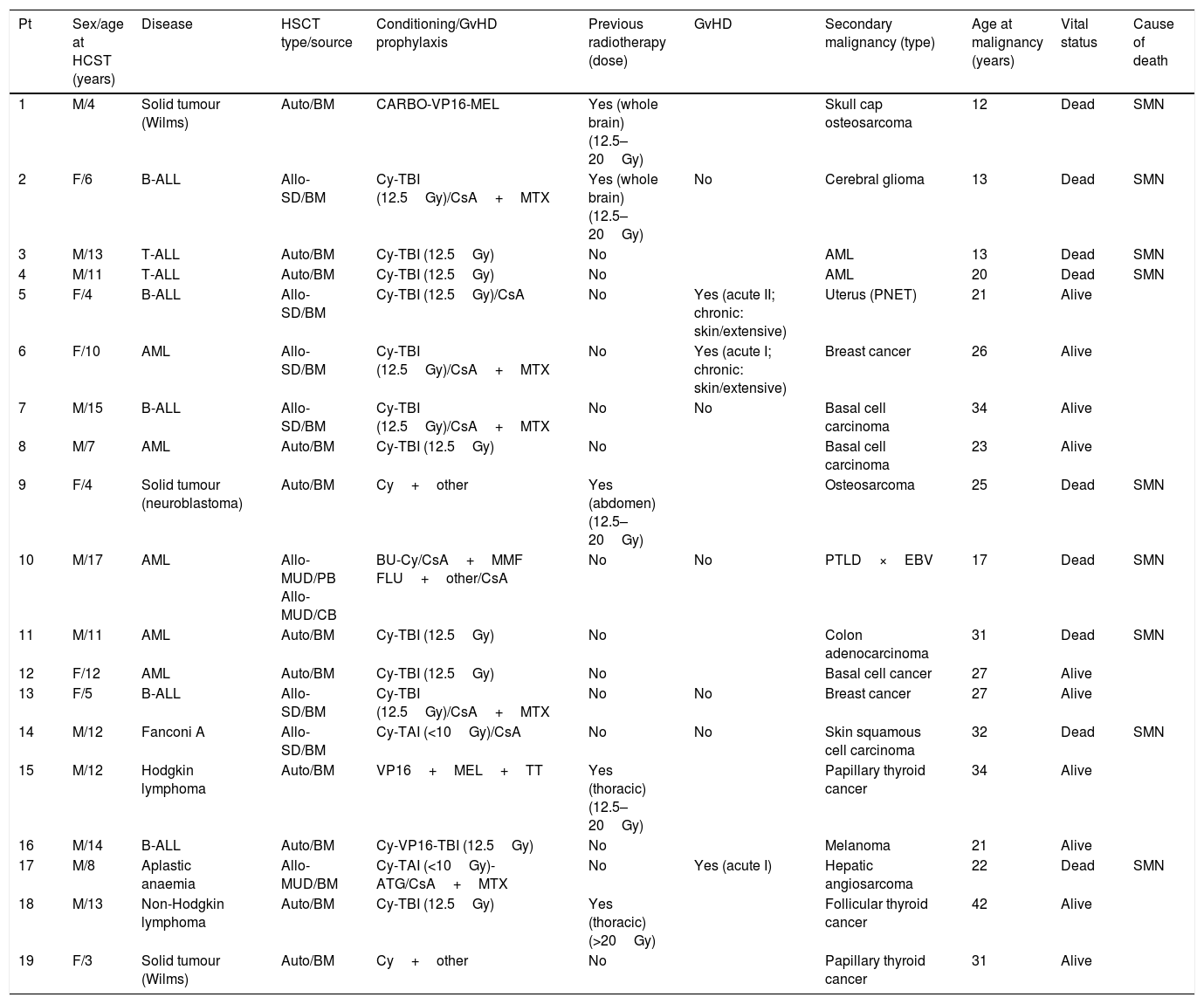
Table 2 summarizes the characteristics of the patients, the HSCTs and the SMNs.
Patient and HSCT characteristics.
| Pt | Sex/age at HCST (years) | Disease | HSCT type/source | Conditioning/GvHD prophylaxis | Previous radiotherapy (dose) | GvHD | Secondary malignancy (type) | Age at malignancy (years) | Vital status | Cause of death |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M/4 | Solid tumour (Wilms) | Auto/BM | CARBO-VP16-MEL | Yes (whole brain) (12.5–20Gy) | Skull cap osteosarcoma | 12 | Dead | SMN | |
| 2 | F/6 | B-ALL | Allo-SD/BM | Cy-TBI (12.5Gy)/CsA+MTX | Yes (whole brain) (12.5–20Gy) | No | Cerebral glioma | 13 | Dead | SMN |
| 3 | M/13 | T-ALL | Auto/BM | Cy-TBI (12.5Gy) | No | AML | 13 | Dead | SMN | |
| 4 | M/11 | T-ALL | Auto/BM | Cy-TBI (12.5Gy) | No | AML | 20 | Dead | SMN | |
| 5 | F/4 | B-ALL | Allo-SD/BM | Cy-TBI (12.5Gy)/CsA | No | Yes (acute II; chronic: skin/extensive) | Uterus (PNET) | 21 | Alive | |
| 6 | F/10 | AML | Allo-SD/BM | Cy-TBI (12.5Gy)/CsA+MTX | No | Yes (acute I; chronic: skin/extensive) | Breast cancer | 26 | Alive | |
| 7 | M/15 | B-ALL | Allo-SD/BM | Cy-TBI (12.5Gy)/CsA+MTX | No | No | Basal cell carcinoma | 34 | Alive | |
| 8 | M/7 | AML | Auto/BM | Cy-TBI (12.5Gy) | No | Basal cell carcinoma | 23 | Alive | ||
| 9 | F/4 | Solid tumour (neuroblastoma) | Auto/BM | Cy+other | Yes (abdomen) (12.5–20Gy) | Osteosarcoma | 25 | Dead | SMN | |
| 10 | M/17 | AML | Allo-MUD/PB Allo-MUD/CB | BU-Cy/CsA+MMF FLU+other/CsA | No | No | PTLD×EBV | 17 | Dead | SMN |
| 11 | M/11 | AML | Auto/BM | Cy-TBI (12.5Gy) | No | Colon adenocarcinoma | 31 | Dead | SMN | |
| 12 | F/12 | AML | Auto/BM | Cy-TBI (12.5Gy) | No | Basal cell cancer | 27 | Alive | ||
| 13 | F/5 | B-ALL | Allo-SD/BM | Cy-TBI (12.5Gy)/CsA+MTX | No | No | Breast cancer | 27 | Alive | |
| 14 | M/12 | Fanconi A | Allo-SD/BM | Cy-TAI (<10Gy)/CsA | No | No | Skin squamous cell carcinoma | 32 | Dead | SMN |
| 15 | M/12 | Hodgkin lymphoma | Auto/BM | VP16+MEL+TT | Yes (thoracic) (12.5–20Gy) | Papillary thyroid cancer | 34 | Alive | ||
| 16 | M/14 | B-ALL | Auto/BM | Cy-VP16-TBI (12.5Gy) | No | Melanoma | 21 | Alive | ||
| 17 | M/8 | Aplastic anaemia | Allo-MUD/BM | Cy-TAI (<10Gy)-ATG/CsA+MTX | No | Yes (acute I) | Hepatic angiosarcoma | 22 | Dead | SMN |
| 18 | M/13 | Non-Hodgkin lymphoma | Auto/BM | Cy-TBI (12.5Gy) | Yes (thoracic) (>20Gy) | Follicular thyroid cancer | 42 | Alive | ||
| 19 | F/3 | Solid tumour (Wilms) | Auto/BM | Cy+other | No | Papillary thyroid cancer | 31 | Alive |
CARBO: carboplatin; MEL: melphalan; Cy: cyclophosphamide; TBI: total body irradiation; CsA: cyclosporine A; MTX: methotrexate; MMF: mycophenolate mofetil; BM: bone marrow; PB: peripheral blood; UCB: umbilical cord blood; SD: sibling donor; MUD: matched unrelated donor; TT: thiotepa; TAI: thoracoabdominal irradiation.
In this cohort, 17 out of 19 patients had received irradiation. During the conditioning, 12 patients received TBI (12.5Gy) and 2 patients thoracoabdominal irradiation (5Gy). Two patients that received TBI-based conditioning had also been treated with radiotherapy before: 1 patient received whole brain radiotherapy (19Gy) and 1 patient thoracic irradiation (>20Gy). Three patients received radiotherapy only for treatment of the underlying disease, but not during conditioning: 1 patient whole brain radiotherapy (19Gy), 1 abdominal irradiation (20Gy) and 1 thoracic irradiation (20Gy). The patients that received whole brain radiotherapy developed the late malignancy at a cranial site: 1 osteosarcoma of the skullcap, and 1 high-grade brain glioma. The secondary malignancy was the cause of death in both cases.
The mortality in patients that developed a SMN was 47.4% (n=9). In all patients that died, the cause was the secondary malignancy.
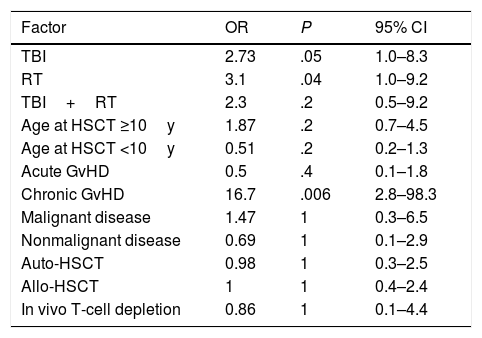
In the univariate analysis, TBI-based conditioning was associated with an increased risk of SMN, although the association was not statistically significant (OR=2.73; P=.05). Previous RT was associated with a 3-fold increase in risk that was statistically significant (OR=.1; P=.04). Chronic GvHD was significantly associated with an increased risk of malignancy (OR=16; P=.006) (Table 3).
Risk factors for subsequent malignancies (univariate analysis).
| Factor | OR | P | 95% CI |
|---|---|---|---|
| TBI | 2.73 | .05 | 1.0–8.3 |
| RT | 3.1 | .04 | 1.0–9.2 |
| TBI+RT | 2.3 | .2 | 0.5–9.2 |
| Age at HSCT ≥10y | 1.87 | .2 | 0.7–4.5 |
| Age at HSCT <10y | 0.51 | .2 | 0.2–1.3 |
| Acute GvHD | 0.5 | .4 | 0.1–1.8 |
| Chronic GvHD | 16.7 | .006 | 2.8–98.3 |
| Malignant disease | 1.47 | 1 | 0.3–6.5 |
| Nonmalignant disease | 0.69 | 1 | 0.1–2.9 |
| Auto-HSCT | 0.98 | 1 | 0.3–2.5 |
| Allo-HSCT | 1 | 1 | 0.4–2.4 |
| In vivo T-cell depletion | 0.86 | 1 | 0.1–4.4 |
TBI: total body irradiation; RT: radiotherapy; GvHD: graft-versus-host disease; OR: odds ratio; 95% CI: 95% confidential interval for odds ratio.
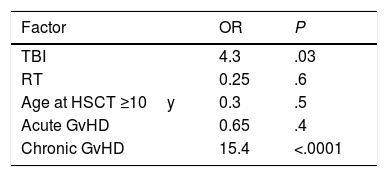
In the multivariate analysis, TBI and chronic GvHD were the factors significantly associated with an increased risk of malignancy. TBI increased the risk by four (OR=4.3; P=.03) and chronic GVHD by more than fifteen (OR=15.4; P<.0001). Previous RT was not associated with an increase in risk in the multivariate analysis (Table 4).
Risk factors for subsequent malignancies (multivariate analysis).
| Factor | OR | P |
|---|---|---|
| TBI | 4.3 | .03 |
| RT | 0.25 | .6 |
| Age at HSCT ≥10y | 0.3 | .5 |
| Acute GvHD | 0.65 | .4 |
| Chronic GvHD | 15.4 | <.0001 |
TBI: total body irradiation; RT: radiotherapy; GvHD: graft-versus-host disease; OR: odds ratio; 95% CI: 95% confidential interval for odds ratio.
In our series, acute GvHD was not associated with an increased risk of SMN. We analysed other factors such as the type of HSCT, the type of underlying disease, age at HSCT, and in vivo depletion of T-lymphocytes, but none of them was associated with an increase in risk.
To verify that malignancies in our cohort were more frequent compared to general population, we compared the incidence in our cohort with the expected incidence in the general population for each age group. The limited number of cases in our study did not allow us to perform a statistical analysis that strictly fulfilled the conditions required for the application of the test. Nevertheless, we confirmed that the incidence was much higher than expected in our cohort, for each malignancy and in male as well as female patients, and that the difference was statistically significant (P<.0001).
We obtained the population data we used for this analysis from the Spanish Cancer Registries Network (Registre de Càncer de Tarragona, Fundació Lliga per a la Investigació i Prevenció del Càncer).
DiscussionSeveral studies have reported that survivors of HSCT have an increased risk of secondary malignant neoplasms, which are an important cause of late mortality.
The cumulative incidence in our study (6%, 12%, and 36% at 15, 20, and 30 years, respectively) confirms that the risk of SMN increases with time. Interestingly, although our data come from a single-centre cohort, the proportions we observed are among the highest of those published. For solid tumours, the reported incidence in the largest series in the literature (for more than 19000 patients) is 1%–6% at 10 years and 2%–15% at 15 years.1 The study published by the researchers of the Center for International Blood and Marrow Transplant Research (CIBMTR) and the Fred Hutchinson Cancer Research Center (FHCRC) reported 189 cases of secondary solid malignancies in a cohort of 28874 allogeneic HSCT recipients (adults and children).3 The cumulative incidence was 1% at 10 years, 2.2% at 15 years and 3.3% at 20 years after HSCT. A European study with a cohort of 1036 patients found a cumulative incidence of solid tumours of 3.5% at 10 years and 12.8% at 15 years.4
In our case series, the most frequent secondary solid tumours were of the thyroid, skin and breast.
Thyroid cancer is a typical radio-induced malignancy that is observed mainly in previously irradiated patients.5,6 However, it has also been described as a secondary tumour after HSCT in patients that have never received irradiation, which suggests that other mechanisms may be at play in the development of these tumours.7 Consistent with these findings, we also found a case of papillary carcinoma in a patient with no history of irradiation.
The association between radiotherapy and an increased risk of basal cell carcinoma (BCC) is well known. Indeed, the basal membrane of epidermis, where 70% of proliferative cells are located, is the most radiosensitive part of the skin.8 A Japanese study of survivors of the atomic bomb underscored that survivors were at increased risk of BCC compared to individuals who had not been exposed to ionizing radiation, with a relative risk of 1.8% (90% CI, 0.8–.3), and that this risk increased with time.9 Skin cancers (non-melanoma type) typically develop in old age, with a median age of onset of 64 years.10 Survivors of HSCT, however, develop skin malignancies at a younger age compared to the general population, usually by 30 years of age, as demonstrated by Perkins et al.10 In our cohort, the 3 patients that developed basal cell carcinomas were aged 23, 27, and 34 years, which confirmed the comparatively earlier onset.
We found two cases of breast cancer that developed before age 30 years. Early age at transplantation and TBI-based conditioning have been described as the main risk factors in the literature. A collaborative study of the FHCRC and the EBMT published in 2008 evaluated the incidence of secondary breast cancer in survivors of allogeneic HSCT.11 This study found 52 cases of cancer in its cohort of 3337 women (1026 of whom had undergone transplant at a paediatric age). The cumulative incidence increased with time: 0.8% at 10 years, 4.6% at 20 years, and 11% at 25 years from HSCT.
In the literature, secondary AML is related to high-dose chemotherapy and autologous HSCT; development of leukaemia from donor cells in survivors of allogeneic HSCTs is extremely rare.12 A study in lymphoma patients with a minimum duration of follow-up of 5 years found a risk for developing AML after autologous HSCT of 3%–19.8%.13 Similarly, the 2 patients that developed AML in our cohort had undergone an autologous HSCT with a TBI-based conditioning regimen.
When it comes to PTLD, the reported incidence in large-scale series is very low (1%–1.6%), and it is generally associated with a poor prognosis.14 Two main risk factors have been identified: intensive immunosuppression, and, in many cases, Epstein–Barr virus (EBV) reactivation.15 In the patient in our cohort, PTLD was associated with EBV reactivation.
One of the objectives of our study was to highlight the risk factors associated with developing a secondary malignancy. Our study corroborated that radiotherapy is one of the main risk factors for SMNs. Interestingly, chronic GvHD was also a major risk factor for SMNs (RR=15.4). In the literature, chronic GvHD has been associated with an increased risk of squamous cutaneous malignancies, mainly in the oral cavity and skin. Some series suggest that immunosuppressive therapy for management of chronic GVHD (such as cyclosporine or azathioprine) is a risk factor for SMNs.16 Chronic inflammation and long-term immunosuppressive therapy can interfere with tissue repair, increasing the risk of developing tumours.
Our results corroborate the role played by irradiation in the development of SMN. Total body irradiation (associated with cyclophosphamide) has been the gold standard for conditioning before HSCT for many years. The shift towards less toxic regimens without irradiation in the last decade may have the benefit of reducing the incidence of late complications such as SMN. We need more time of follow-up to analyse the impact of these novel reduced-intensity conditioning regimens on the development of secondary tumours. Another important aspect corroborated by our data is that chronic GvHD is a complication that seems to carry an increased risk of SMN. Chronic GvHD prophylaxis thus appears to be the best available strategy to prevent late malignancies. However, its use must be carefully weighed against the associated increase in disease relapse in patients in whom HSCT was performed to treat malignant disease.
The high incidence of late malignancies highlights the need for life-long surveillance of HSCT survivors.
One of the main strengths of our study was the long duration of follow-up, as the incidence of secondary malignancies tends to increase with time, and it allowed us to report several cases of SMN despite the small size of the study cohort. In addition, there were no losses to follow-up, so we were able to report the actual incidence of SMNs. There are also limitations to our study. Despite the high number of SMNs we observed, the small size of our cohort did not allow us to reach generalizable conclusions. The heterogeneity in chemotherapy treatments for solid tumours and leukaemia prevented us from analysing the impact of these previous treatments on the risk of secondary malignancies. Lastly, we did not study the possible role in oncogenesis of other exogenous factors (alcohol, tobacco, obesity, hormonal treatment, etc.) or genetic factors.
In conclusion, subsequent malignant neoplasms constitute an important cause of morbidity and mortality in long-term survivors of hematopoietic stem cell transplantation. We have observed a very high incidence of SMNs in patients that underwent HSCT in the paediatric age. Radiotherapy and chronic GvHD were the main risk factors associated with the development of SMN in our study.
Conflicts of interestThe authors declare that they have no conflicts of interest.
We want to thank Jaume Galceran, head of the Spanish Cancer Registries Network, Registre de Càncer de Tarragona, Fundació Lliga per a la Investigació i Prevenció del Càncer, Reus (Tarragona, Spain).
Please cite this article as: Sisinni L, Gich I, Torrent M, Badell I. Neoplasias malignas secundarias después de un trasplante de progenitores hematopoyéticos en edad pediátrica. An Pediatr (Barc). 2019;90:157–164.