Food allergy is an increasing health problem in the developed world. Cow's milk protein is the main cause of food allergy in infants. Without an appropriate diagnostic workup, there is a high risk of both over- and underdiagnosis and therefore, over and undertreatment. The objective of our study was to analyze the variability in cow's milk protein allergy (CMPA) management by pediatric gastroenterologists in Spain.
MethodsA 50 item questionnaire, including open and closed items in a Likert's scale from 0 to 5, was drafted and distributed through the Spanish Society for Pediatric Gastroenterology, Hepatology and Nutrition (SEGHNP) e-mail list.
ResultsSeventy-three questionnaires were received back out of 321. Only 3 of the items achieved concordance greater than 90%. Thirty-three percent considered oral challenge to be necessary for the diagnosis of CMPA under any circumstance. Twenty-five percent considered that symptom improvement after cow's milk removal was enough for the diagnosis. Oral challenge was performed at home by 83.5% in non-IgE mediated cases. Extensively hydrolyzed casein formulas were the treatment of choice for 69.9%. Soy formulas were the last option. Almost all respondents were aware of the existence of clinical guidelines on CMPA, being European Society of Pediatric Gastroenterology, Hepatology and Nutrition guidelines the most followed (64.4%). Twenty-three percent considered that their knowledge about allergy was inadequate.
ConclusionsAlthough CMPA is a prevalent condition that pediatric gastroenterologists have been treating for decades, we found a huge variability on its management. There is potential for improvement in this field among pediatric gastroenterologist in the future.
La alergia alimentaria es un problema creciente, siendo la proteína de leche de vaca la principal causa en niños. Sin un proceso diagnóstico adecuado, existe un elevado riesgo de sobrediagnóstico e infradiagnóstico y, por lo tanto, de sobretratamiento e infratratamiento. El objetivo de nuestro estudio fue analizar la variabilidad en el manejo de la alergia a proteína de leche de vaca (APLV) por los gastroenterólogos pediátricos españoles.
MétodosSe envió un cuestionario de 50 preguntas a través de la lista de email de la Sociedad Española de Gastroenterología, Hepatología y Nutrición Pediátricas.
ResultadosRecibimos 73 cuestionarios de los 321 enviados. Solo 3 de las respuestas lograron más del 90% de acuerdo. El 33% considera que la provocación oral es necesaria para el diagnóstico de APLV siempre. El 25% considera que la mejoría clínica tras la retirada de las proteínas de leche de vaca es suficiente para el diagnóstico. La provocación oral es realizada en domicilio por el 83,5% de los encuestados en APLV no IgE mediada. Los hidrolizados extensos de caseína son el tratamiento de elección (69,9%). Las fórmulas de soja, la última opción. Casi todos los encuestados conocían la existencia de guías de manejo de APLV, siendo las de la Sociedad Europea de Gastroenterología, Hepatología y Nutrición Pediátrica las más utilizadas (64,4%). El 23% considera que su conocimiento sobre alergia es inadecuado.
ConclusionesAunque la APLV es una patología prevalente que los gastroenterólogos pediátricos llevan décadas tratando, hemos encontrado una gran variabilidad en su manejo. Existe posibilidad de mejora en este campo en el futuro.
Food allergy is an increasing health care concern. Food allergy is defined as an adverse health effect secondary to a specific immune response that occurs reproducibly following exposure to a given food. The immune reaction may be IgE mediated, non-IgE mediated, or mixed. Cow's-milk protein is the leading cause of food allergy in infants and young children.1 Without an appropriate diagnostic workup, there is a high risk of both over- and underdiagnosis and therefore, over and undertreatment. This could affect dramatically to children's quality of life. Moreover, milk avoidance, when it is not necessary, may disturb child's nutritional status or growth development.2,3
Although cow's milk protein allergy (CMPA) is a prevalent condition, its management is quite different depending on the attending physician. Several guidelines4–7 from different organizations have been recently published in highly read journals, trying to unify the management of this condition among pediatric practitioners involved, including primary care pediatricians, pediatric gastroenterologists and pediatric allergologists.
The aim of our study was to analyze the variability in CMPA management by different pediatric gastroenterologist divisions in Spain.
MethodsA fifty-item questionnaire (Appendix A) was composed and sent through the Spanish Society for Pediatric Gastroenterology, Hepatology and Nutrition (SEGHNP) e-mail list, where the vast majority of pediatric gastroenterologists in Spain are included. This Society has a total of 321 members, among which 121 of them are full members. The questionnaire comprised open and closed items in a Likert's scale from 0 to 5, including questions about CMPA diagnosis, treatment and prognosis. The items score were grouped as follows: 0–1 “disagree”; 2–3 “undefined”; 4–5 “agree”. The questionnaire was sent January 15th 2016 and the study was closed on January 31st. Data were anonymously included in a database and then analyzed with SPSS 20.0.
ResultsSeventy-three questionnaires were received back. 56.9% of the respondents were SEGHNP full members. The median time of professional experience of the participants, was 16 years, ranging from 5 to 43 years.
Only 3 of the items reached more than 90% of concordance among the survey respondents. In other words, for those 3 items more than 90% of the survey respondents agreed with the same answer. In 11 items, the median score fell into the undefined category (2–3 points).
More than 90% of the survey respondents disagreed with the statement that a negative specific IgE test, ruled out CMPA. On the contrary, a 90% agreement was observed with the two following items: after a positive oral challenge, it is mandatory to avoid CMP during, at least, 6 months and by 2 years of age, 80% of CMPA infants will develop tolerance to CMP.
A huge variability was observed regarding answers to the open label questions so those data are not shown in the results.
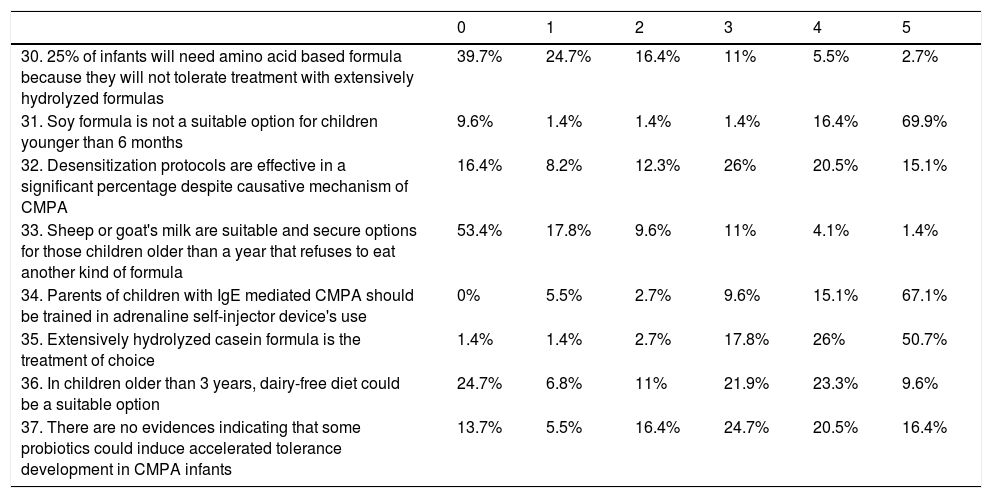
DiagnosisAccording to diagnosis (Table 1), 25% of the participants answered that symptoms relief after CMP removal was enough to diagnose CMPA. Forty-three percent of the respondents considered that Oral Challenge (OC) should be performed always in a hospital setting but when they were specifically asked about mild non-IgE mediated CMPA cases, 83.5% agreed with the possibility of reintroducing milk at home.
Diagnosis.
| 0 | 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|---|
| 5. Cow's milk protein allergy (CMPA) diagnosis is based mainly on the positivity of any specific IgE test | 60.3% | 13.7% | 11% | 8.2% | 4.1% | 1.4% |
| 6. If specific IgE determination in blood is negative, any kind of CMPA could be ruled out | 86.3% | 6.8% | 4.1% | 0% | 1.4% | 1.4% |
| 7. If casein specific IgE levels are positive, oral food challenge (OFC) should not be performed independent on their level in blood | 71.2% | 16.4% | 1.4% | 5.5% | 2.7% | 1.4% |
| 8. OFC could be avoided under certain circumstances | 5.5% | 1.4% | 6.8% | 11% | 15.1% | 58.9% |
| 9. OFC is mandatory for CMPA diagnosis | 19.2% | 13.7% | 9.6% | 20.5% | 21.9% | 13.7% |
| 10. Symptoms improvement after milk's withdrawal is enough most of the times to establish CMPA diagnosis | 28.8% | 11% | 17.8% | 17.8% | 15.1% | 9.6% |
| 11. Non IgE mediated CMPA diagnosis is based mainly on the positivity of a patch test | 69.9% | 16.4% | 8.2% | 2.7% | 2.7% | 0% |
| 12. To evaluate efficacy in a diagnostic elimination diet, milk's withdrawal should be maintained for at least a month | 8.2% | 5.5% | 15.1% | 20.5% | 27.4% | 23.3% |
| 13. OFC cannot be avoided under any circumstance | 54.8% | 5.5% | 15.1% | 11% | 8.2% | 4.1% |
| 14. After a positive OFC, infants must be on a milk-free diet for at least 6 months | 2.7% | 1.4% | 5.5% | 26% | 63% | 98.6% |
| 15. After a positive OFC, any subsequent challenge should be delayed after the age of 12 months | 12.3% | 13.7% | 9.6% | 17.8% | 19.2% | 27.4% |
| 16. Oral food challenge must always be performed in a hospital setting | 21.9% | 9.6% | 5.5% | 17.8% | 15.1% | 28.8% |
| 17. In a patient presenting with perioral rash after every exposition to cow's milk proteins, in which symptoms improve after cow's milk withdrawal, if specific IgE test are negative, a diagnosis of non IgE CMPA could be made | 30.1% | 11% | 9.6% | 12.3% | 13.7% | 21.9% |
| 18. In patients with negative specific IgE in which CMPA is still suspected, and the determination of a specific IgG to CMP is positive, it's mandatory to remove milk from the infants’ diet | 52.1% | 13.7% | 12.3% | 9.6% | 5.5% | 6.8% |
The questionnaire comprised open and closed items in a Likert's scale from 0 to 5, including questions about CMPA diagnosis, treatment and prognosis. The items score were grouped as follows: 0–1 “disagree”; 2–3 “undefined”; 4–5 “agree”. The following table summarizes the answers (expressed in percentage) related to diagnosis of CMPA (Questions 5 to 18).
Related to the diagnostic workup, 46.5% recognized routine use of the patch test and 50% always performed both prick test and blood sampling for specific IgE antibodies. OC was mandatory to diagnose CMPA for 36% of the respondents. Moreover, 12% considered that it could never be avoided. OC protocols were almost different in every unit and were performed equally by pediatric allergologists and gastroenterologists.
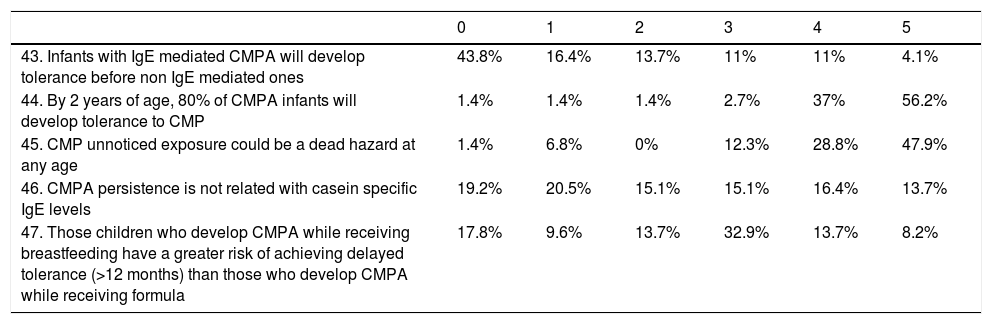
TreatmentAccording to treatment (Table 2), 11% thought that soy formula was an appropriate treatment for children under 6 months of age, and 5.5% believed that milk from other mammals was a suitable alternative option. A majority of the respondents (67.1%) did not consider a milk free diet in their protocols in children older than 3 years.
Treatment.
| 0 | 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|---|
| 30. 25% of infants will need amino acid based formula because they will not tolerate treatment with extensively hydrolyzed formulas | 39.7% | 24.7% | 16.4% | 11% | 5.5% | 2.7% |
| 31. Soy formula is not a suitable option for children younger than 6 months | 9.6% | 1.4% | 1.4% | 1.4% | 16.4% | 69.9% |
| 32. Desensitization protocols are effective in a significant percentage despite causative mechanism of CMPA | 16.4% | 8.2% | 12.3% | 26% | 20.5% | 15.1% |
| 33. Sheep or goat's milk are suitable and secure options for those children older than a year that refuses to eat another kind of formula | 53.4% | 17.8% | 9.6% | 11% | 4.1% | 1.4% |
| 34. Parents of children with IgE mediated CMPA should be trained in adrenaline self-injector device's use | 0% | 5.5% | 2.7% | 9.6% | 15.1% | 67.1% |
| 35. Extensively hydrolyzed casein formula is the treatment of choice | 1.4% | 1.4% | 2.7% | 17.8% | 26% | 50.7% |
| 36. In children older than 3 years, dairy-free diet could be a suitable option | 24.7% | 6.8% | 11% | 21.9% | 23.3% | 9.6% |
| 37. There are no evidences indicating that some probiotics could induce accelerated tolerance development in CMPA infants | 13.7% | 5.5% | 16.4% | 24.7% | 20.5% | 16.4% |
The questionnaire comprised open and closed items in a Likert's scale from 0 to 5, including questions about CMPA diagnosis, treatment and prognosis. The items score were grouped as follows: 0–1 “disagree”; 2–3 “undefined”; 4–5 “agree”.
The following table summarizes the answers (expressed in percentage) related to treatment of CMPA (Questions 30 to 37).
When they were asked about their formula of choice, a 69.9 percent indicated extensively hydrolyzed casein formulas as their first option. Extensively hydrolyzed whey formulas were the second option for 50.7%. The third choice was amino acid based formulas for 39.7%. As a fourth option 46.6% chose hydrolyzed rice formula. Soy formulas were chosen as the last option in 43.8% of the cases.
Although 79.4% had desensitization protocols in their divisions, nearly all units followed a different protocol. Sixty three percent had protocols for IgE mediated CMPA and 16.4% for both IgE and non IgE mediated CMPA.
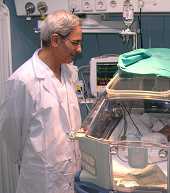
PrognosisRelated to prognosis (Table 3), 8.2% of the respondents did not consider CMP exposition as a possible death hazard at any age. Twenty four percent of the respondents were not sure if non-IgE mediated CMPA infants developed tolerance before or after IgE mediated ones.
Prognosis.
| 0 | 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|---|
| 43. Infants with IgE mediated CMPA will develop tolerance before non IgE mediated ones | 43.8% | 16.4% | 13.7% | 11% | 11% | 4.1% |
| 44. By 2 years of age, 80% of CMPA infants will develop tolerance to CMP | 1.4% | 1.4% | 1.4% | 2.7% | 37% | 56.2% |
| 45. CMP unnoticed exposure could be a dead hazard at any age | 1.4% | 6.8% | 0% | 12.3% | 28.8% | 47.9% |
| 46. CMPA persistence is not related with casein specific IgE levels | 19.2% | 20.5% | 15.1% | 15.1% | 16.4% | 13.7% |
| 47. Those children who develop CMPA while receiving breastfeeding have a greater risk of achieving delayed tolerance (>12 months) than those who develop CMPA while receiving formula | 17.8% | 9.6% | 13.7% | 32.9% | 13.7% | 8.2% |
The questionnaire comprised open and closed items in a Likert's scale from 0 to 5, including questions about CMPA diagnosis, treatment and prognosis. The items score were grouped as follows: 0–1 “disagree”; 2–3 “undefined”; 4–5 “agree”.
The following table summarizes the answers (expressed in percentage) related to prognosis of CMPA (Questions 43 to 47).
European Society of Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) guidelines were the most widely used, with a 64% of the respondents following those guidelines. All the respondents were aware of the existence of published guidelines, but 23% of them considered that their knowledge on CMPA was outdated and scarce.
DiscussionAlthough CMPA is a very prevalent condition which pediatric gastroenterologists have been treating for decades, there is still huge variability on its management. Without an appropriate diagnostic workup, including food challenge procedures, there is a high risk of both over- and under-diagnosis. In consequence, we are under risk of over- and under-treatment.
We have observed a huge variability among pediatric gastroenterologists in Spain regarding CMPA diagnosis and management. Not surprisingly, the vast majority of the pediatric gastroenterologists in Spain, were aware of the existence of clinical practice guidelines on the problem, and most of them acknowledged to follow the ESPGHAN guidelines, published in 2012.1 Nevertheless, significant differences in clinical practice were found compared to the ESPGHAN recommendations both in diagnosis and treatment.
ESPGHAN guidelines indicate clearly when and when not to perform an OC in patients suspected to have CMPA. All cases of non IgE mediated CMPA, with the exception of Food Protein Induced Enterocolitis Syndrome (FPIES) should undergo an OC to be adequately diagnosed. In IgE mediated cases, with a clear reaction close to the moment of milk ingestion and with a positive specific IgE testing to CMP, the OC could be avoided. In our survey, 36% participants considered OC to be mandatory always. One in each four pediatric gastroenterologists considered symptom resolution on CMP exclusion diet was enough to diagnose CMPA.
A high percentage of respondents considered that exposition to milk could be performed at home in non severe, non IgE mediated CMPA patients, a scenario considered in the ESPGHAN guidelines. When asked to describe the protocol used for milk reintroduction at home, there were almost no coincidences, being nearly one different protocol per unit in the whole country.
Nearly half of the participants reported regular use of patch tests in their offices for the diagnosis of CMPA, a slightly higher percentage than reported in the literature.8 However, 76% of the respondents thought that non IgE mediated CMPA diagnosis should not be made exclusively based on its results. In current ESPGHAN guidelines, these tests are not recommended. The patch test is difficult to read and subjective. Moreover, there are difficulties on its standardization and application of antigen. In any case, recent studies indicate that they are reliable and safe but its use cannot substitute oral challenge in most of the cases.9
Only 11% of the respondents considered soy formula to be an adequate option for infants below six months of age. ESPGHAN discourages soy formula consumption in this age group because of its phytates and isoflavones content. Not surprisingly, only 5.5% considered milk from other mammals as a suitable option in the treatment of CMPA infants. This option should always be avoided8 because cow, goat and sheep milks evidence 87–98% identity of amino acids and immunological cross reactivity is a real issue.
In children older than 3 years of age, a diet with no dairy content is considered to be a safe option for treatment if CMPA persists beyond that age, given that calcium and vitamin D are adequately supplied in the child's diet. In our survey, close to 70% of the respondents did not consider this as a safe option. In the Spanish National Health System, dietitians are scarce. Those pediatric gastroenterologists, with no help from experienced pediatric dietitians, could feel insecure to recommend this diet to children under these circumstances.10
Although most of the CMPA infants will outgrow their allergy before the age of three, a significant proportion of children will continue to be allergic to cow's milk, being this percentage higher for those with an IgE mediated allergy.11 Four out of 10 of the respondents were not sure if this statement was true. In the last years, oral desensitization protocols (OTI) have been developed for these IgE patients and are widely used, even when several concerns have been raised regarding the possibility of developing eosinophilic esophagitis during the treatment. In our study, almost 90% of the respondents have access to these protocols in their practices.12
There are several reports indicating that accidental exposure to cow's milk has been responsible of deaths, and all CMPA children with an anaphylactic reaction or at risk of developing one and their parents should be instructed in the use of epinephrine injectors for that cause. Close to 10 percent of the responders to our survey, did not recognize accidental exposure to milk as a hazardous risk at any age.13,14
Finally, one out of four pediatric gastroenterologists in Spain, considered that their knowledge on CMPA was outdated and scarce, even when almost all of them were aware of the existence of practice guidelines. Continued medical education systems are not always capable of maintaining practitioners updated. Several initiatives have been conducted in order to enhance medical knowledge on food allergy management. Yu et al., carried out a study that developed and piloted a food allergy educational resource with very good results. Self-paced online training modules were selected by their survey respondent as one of the most preferred learning methods. Based upon this, the educational material comprised a slide set and written material as well as a self-injectable epinephrine demonstration. Significant improvement in knowledge and confident was observed after completed pre-and post-knowledge test.15
Another brief educational tool, is the Food Allergy Comprehension Tool (FACT) that is a rapid way to address known knowledge gaps among pediatricians and to identify areas in need of further intervention. It was designed to integrate with the current food allergy guidelines. It includes a pre- and post-evaluation, allergy case studies and several case-related questions with detailed explanations. Significant improvements in knowledge and confidence were observed.16
We have found surveys regarding CMPA management focused on primary care physicians. The goal of a survey carried by Gupta and coworkers,17 was to characterize food allergy knowledge, attitudes, and beliefs among primary care physicians and family physicians in the United States. Participants answered 61% of knowledge based items correctly. The overall score was significantly higher for pediatricians (62%) than for family physicians (54%).
A Spanish study18 found low awareness of existing guidelines for CMA among general pediatricians, pediatric nutritionists and allergologists. 39% of their survey respondent from 52 countries were unaware of CMA management guidelines. They also found a potential over-diagnosis among general pediatricians in Europe due to variable diagnostic practices.
To the best of our knowledge, this is the first survey conducted among pediatric gastroenterologists on this topic.
The strengths of our study were that in Spain, nearly all non-IgE mediated and many IgE mediated CMPA patients are managed by pediatric gastroenterologists, and the vast majority of them were reached through the mailing list. Twenty-three percent of the Spanish pediatric gastroenterologists answered the survey with a high percentage of full members, and data were collected and analyzed in a short period of time, avoiding communication among participants. The median time of professional experience was over 15 years, and more than half of the respondents were full members, indicating that participants in the study are experienced professionals with a high degree of specialization.
The main limitation of the study is a possible selection bias in the participants. Data were anonymously included in a database, but we can assume that respondents were those more confident in their answers or with a greater interest in CMPA, so we could reasonably expect to get better results in this group. It may well be that the final results may underestimate the real magnitude of the problem.
In conclusion, although CMPA is a prevalent condition that pediatric gastroenterologists have been treating for decades, we have found huge variability on its management. There are multiple opportunities for improvement in CMPA diagnostic workup and management among pediatric gastroenterologist in the future.
Conflict of interestThe authors declare that they have no conflict of interest.
Thank you to all the participants in our survey.
The following questionnaire is anonymous. It comprises 50 open and Likert's scale questions. It requires 15min approximately to be completed.
Thank you in advance for your cooperation.
Identity data
- 1.
Gender:
- 2.
Age:
- 3.
Type of member (full members/associate members):
- 4.
Years of experience (fellowship included):
Diagnosis
Please note your agreement with the following statements (from 0=no agreement at all to 5=complete agreement)
- 5.
Cow's milk protein allergy (CMPA) diagnosis is based mainly on the positivity of any specific IgE test.
- 6.
If specific IgE determination in blood is negative, any kind of CMPA could be ruled out.
- 7.
If casein specific IgE levels are positive, oral food challenge (OFC) should not be performed independent on their level in blood
- 8.
OFC could be avoided under certain circumstances.
- 9.
OFC is mandatory for CMPA diagnosis.
- 10.
Symptoms improvement after milk's withdrawal is enough most of the times to establish CMPA diagnosis.
- 11.
Non IgE mediated CMPA diagnosis is based mainly on the positivity of a patch test.
- 12.
To evaluate efficacy in a diagnostic elimination diet, milk's withdrawal should be maintained for at least a month.
- 13.
OFC cannot be avoided under any circumstance.
- 14.
After a positive OFC, infants must be on a milk-free diet for at least 6 months.
- 15.
After a positive OFC, any subsequent challenge should be delayed after the age of 12 months.
- 16.
Oral food challenge must always be performed in a hospital setting
- 17.
In a patient presenting with perioral rash after repeated exposition to cow's milk proteins, in which symptoms improve after cow's milk withdrawal, if specific IgE tests are negative, a diagnosis of non IgE CMPA could be made.
- 18.
In patients with negative specific IgE in which CMPA is still suspected, and the determination of a specific IgG to CMP is positive, it is mandatory to remove milk from the infants’ diet.
The following questions are meant to be answered freely:
- 19.
In a patient with a suspected CMPA. In which circumstance you won’t carry out an IgE blood sample/prick test?
- 20.
If you have a patient with a suspected IgE mediated CMPA, Which test will you ask for: prick test, IgE specific blood sample or both?
- 21.
Are patch tests carried out at your Hospital?
- 22.
Who performs oral challenges at your Hospital? Gastroenterologist or allergologist?
- 23.
Describe the protocol followed to do it:
- 24.
In patients older than 24 months of age with persistent IgE mediated CMPA symptoms without a story of anaphylaxis. Would you determine specific IgE prior to the exposure?
- 25.
In the previous situation. Would you carry out an oral food challenge if specific IgE turns out positive? Yes, always/no, never/sometimes.
- 26.
If your answer was “sometimes” please specify when.
- 27.
Do you carry out CMP oral challenges at home?
- 28.
If your answer is yes, please note when:
- 29.
If your answer is yes, please explain how do you perform the exposition.
Treatment
Note your agreement with the following statements (from 0=no agreement at all to 5=complete agreement)
- 30.
25% of infants will need an amino acid based formula because they will not tolerate treatment with extensively hydrolyzed formulas.
- 31.
Soy formula is not a suitable option for children younger than 6 months.
- 32.
Desensitization protocols are effective in a significant percentage despite the causative mechanism of CMPA.
- 33.
Sheep or goat milks are suitable and secure options for those children older than a year that refuse to eat another kind of formula.
- 34.
Parents of children with IgE mediated CMPA should be trained in adrenaline self-injector device's use.
- 35.
Extensively hydrolyzed casein formula is the treatment of choice.
- 36.
In children older than 3 years, dairy-free diet could be a suitable option.
- 37.
There are no evidences indicating that some probiotics could induce accelerated tolerance development in CMPA infants
The following questions are meant to be answered freely:
- 38.
Do you carry out desensitization protocols at your hospital?
- 39.
Which kind of protocol do you carry out? (standard/accelerated).
- 40.
At what age?
- 41.
Do you carry out the protocol in patients with IgE mediated CMPA, non IgE mediated CMPA or both?
- 42.
Please, order the following formulas according to your prescription frequency (from more frequent to less frequent prescription): elemental formula, extensively hydrolyzed seroprotein formulas, extensively hydrolyzed casein formulas, hydrolyzed rice formulas, soy formula.
Prognosis
Please note your agreement with the following statements (from 0=no agreement at all to 5=total agreement)
- 43.
Infants with IgE mediated CMPA will develop tolerance before non IgE mediated ones
- 44.
By 2 years of age, 80% of CMPA infants will develop tolerance to CMP.
- 45.
CMP unnoticed exposure could be a death hazard at any age.
- 46.
CMPA persistence is not related with casein specific IgE levels.
- 47.
Those children who develop CMPA while receiving breast feeding have a greater risk of achieving delayed tolerance (>12 months) than those who develop CMPA while receiving formula.
The following questions are meant to be answered freely:
- 48.
Do you know about the existence of CMPA clinical practice guidelines?
- 49.
If your answer is yes, please note which one is the one you follow in your daily practice.
- 50.
Do you think that your knowledge about CMA is appropriate? (0=nothing appropriate, 5=completely appropriate).
Thank you for your cooperation
Please cite this article as: Pérez AI, Sánchez AM, Cantón ÓS, Jaime BE, Treviño SJ, García CB, et al. Manejo de la alergia a proteína de leche de vaca por los gastroenterólogos españoles. An Pediatr (Barc). 2018;89:222–229.
We presented these results in the Spanish Congress of Pediatric Gastroenterology, Hepatology and Nutrition in Gijón, May 2016 and in the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition in Montreal, October 2016.