To identify risk factors associated with infectious complications acquired by paediatric patients during extracorporeal life support (ECLS).
Patients and methodsPatients under ECLS from January 2011 to December 2014 have been retrospectively reviewed and data on demographics, care and infectious complications were collected.
ResultsThere were 50 ECLS assistances in the study period, of which 20 patients had 23 infectious complications: 16 were bloodstream infections, with coagulase negative staphylococci being the predominant isolate (there were 2 cases of candidaemia). Age, site of cannulation procedure, cannulation site, severe coagulopathy, and surgical interventions during assistance were analysed as risk factors for infectious complications, but no significant differences were found. ECLS duration was significantly longer in patients with infectious complications (8.91 vs 5.91 days; P=.039). There were no significant differences as regards Paediatric Intensive Care Unit (PICU) stay, or in survival.
ConclusionsInfectious complications during ECLS are very common, and ECLS duration is significantly longer in patients with infections. Measures should be put in place to prevent infectious complications and reduce time on ECLS.
Identificar factores de riesgo asociados con el desarrollo de infecciones nosocomiales en pacientes pediátricos asistidos con oxigenación por membrana extracorpórea (ECMO).
Pacientes y métodosSe han revisado de forma retrospectiva los pacientes que han recibido asistencia en ECMO de enero de 2011 a diciembre de 2014. Se han recogido datos demográficos, sobre la asistencia y sobre las infecciones que aparecen durante dicha asistencia.
ResultadosEn este periodo hubo 50 asistencias en ECMO. Veinte pacientes tuvieron 23 episodios de infección, de los que 16 fueron bacteriemias, siendo el microorganismo más frecuente el estafilococo coagulasa negativo (habiendo 2 casos de candidemia). En cuanto a los grupos de edad, el lugar y el tipo de canulación, la presencia de coagulopatía grave y la realización de intervenciones quirúrgicas durante la asistencia, ninguno de estos factores resultó un factor de riesgo de infección estadísticamente significativo. La duración media de soporte en ECMO fue significativamente mayor en los pacientes que tuvieron alguna infección (8,91 vs. 5,96 días; p=0,039). No hubo diferencias significativas en cuanto a la estancia en la Unidad de Cuidados Intensivos Pediátricos ni en cuanto a la supervivencia.
ConclusionesLa incidencia de infección durante ECMO es muy alta y los pacientes que tienen una infección están asistidos un tiempo significativamente mayor, por lo que se deben instaurar medidas para prevenir la aparición de estas infecciones e intentar minimizar el tiempo de asistencia en ECMO.
Extracorporeal membrane oxygenation (ECMO) is a technique used in critical patients of any age with reversible cardiopulmonary failure.1 The development of infectious complications in patients supported by ECMO is associated with poorer outcomes.2,3 Patients under ECMO are particularly susceptible to nosocomial infections, since they not only are critically ill but also undergo numerous invasive procedures (placement of central venous and arterial catheters, urinary catheters, abdominal or chest drainage tubes, endotracheal intubation, etc.).4,5 In addition, the ECMO circuit itself contains several potential points of entry for pathogenic microorganisms.
Several studies have described nosocomial infections in the context of ECMO.3,6 The known risk factors for nosocomial infection include longer duration of ECMO,6,7 greater disease severity at initiation of ECMO,2,7 performance of invasive procedures during ECMO,5 ECMO support with an open chest5 and complications during ECMO.6,8 A query of the Extracorporeal Life Support Organization (ELSO) Registry (1998–2008) found that infections developed in 11.7% of ECMO cases, which corresponds to a rate of 15.4 infections per 1000 ECMO days (30.6/1000 d in adult patients, 20.8/1000 in paediatric patients and 10.1/1000d in neonatal patients); the proportion dropped to 6.1% in patients that required ECMO for 7 or fewer days and rose to 30.3% in patients that required it for more than 14 days.3
The aim of this study was to make a retrospective analysis of the patients that received ECMO support in the paediatric intensive care unit (PICU) of a tertiary care hospital to describe the associated infections, identify the risk factors for infectious complications and find opportunities for improvement.
Patients and methodsWe performed a retrospective review of the health records of patients that received ECMO support between January 1, 2011 and December 31, 2014. We collected data on the demographic characteristics of patients, the care received (indication for ECMO, type of ECMO, type of cannulation, setting of insertion, procedures during ECMO, transfusion of blood products, ECMO duration), the infections developed during ECMO (type of infection, aetiological agent, time from ECMO initiation to onset of infection), length of stay in the PICU (in days) and survival.
The study included all patients aged 0–16 years that received ECMO support during this period. We excluded patients with infections with onset before to initiation of ECMO, that were the indication for ECMO, or that developed after ECMO was discontinued and may not be secondary to this support.
Each type of nosocomial infection was diagnosed based on the criteria established in the CDC guidelines.9 In our hospital, cultures are performed routinely every 48h.
During the period under study, antibiotic prophylaxis in patients that received ECMO consisted of a 48-h course of vancomycin and gentamycin.
We compared continuous variables by means of analysis of variance, and qualitative variables by means of the χ2 test or the Fisher exact test. We expressed the results as percentages and P-values (statistical significance was defined as P<.05). The statistical analysis was performed with SPSS version 19.0 (IBM).
ResultsDuring the period under study, 50 patients required ECMO in our unit. Table 1 details the demographic characteristics, indication for ECMO and characteristics of ECMO for these patients.
Summary of demographic and ECMO characteristics.
| Median age: 2 months (IQR, 0–9.75 months) |
| Median weight: 3.9kg (IQR, 3.100–8.250kg) |
| Sex |
| Male: 54% (27) |
| Female: 46% (23) |
| Indication for ECMO |
| Post cardiac surgery: 58% (29) |
| Severe ventricular failure: 12% (6) |
| Respiratory failure: 18% (9) |
| ECMO-CPR: 12% (6) |
| A-V ECMO: 100% (50) |
| Mean duration of ECMO: 7.26 days (2.36–12.16) |
| Type of cannulation |
| Central: 76% (38) |
| Peripheral: 24% (12) |
| Setting of cannulation |
| Operating theatre: 58% (29) |
| PICU: 42% (21) |
| Procedures during ECMO: 58% (29) (bleeding control, repeat surgery or catheterization) |
| Massive transfusion: 40% (20) |
| Infection during ECMO: 40% (20) |
| Mean time elapsed from ECMO initiation at onset of infection: 4.48 days (1.21–7.75) |
| Mean length of stay in PICU: 39.16 days (1.42–77) |
| Overall survival: 74% (37) |
In this period, 20 patients (40%) had at least 1 infection (3 patients had 2 infections), and the mean time elapsed between ECMO initiation and the onset of infection was 4.48 days (1.21–7.75). There were 23 episodes of infection (16 bloodstream infections, 4 urinary tract infections, 1 case of ventilation-associated pneumonia and 2 cases of mediastinitis). Measurement of C-reactive protein (CRP) and procalcitonin (PCT) levels within 48h of the onset of infection showed that CRP levels peaked at 48h (mean, 12mg/dL; 2.4–21.6) and PCT levels in the first 12h (mean, 10ng/mL; 0–23). Table 2 specifies the causative organism in each infection. When we analysed the microorganisms that caused infection in patients managed with ECMO, we found that gram-positive bacteria were the most frequently involved group of organisms (40%), followed by gram-negative bacteria (35%) and fungi (25%). Coagulase-negative staphylococci were the most frequent causative agent (involved in 6 of the 23 infections), followed by various Enterobacteriaceae (5 out of 23 infections) and Candida spp. (5 out of 23 infections).
Microorganisms that caused infection.
| Bloodstream infection | |||||
|---|---|---|---|---|---|
| Gram-positive | Gram-negative | Fungi | |||
| Coagulase-negative Staphylococcus spp. | 5 | Stenotrophomonas maltophilia | 1 | Candida albicans | 1 |
| Enterococcus faecium | 1 | Klebsiella pneumoniae | 1 | Candida parapsilosis | 1 |
| Streptococcus viridans | 1 | Serratia marcescens | 1 | ||
| Granulicatella adiacens | 1 | Pseudomonas aeruginosa | 2 | ||
| Enterobacter cloacae | 1 | ||||
| Urinary tract infection | |||||
|---|---|---|---|---|---|
| Gram-positive | Gram-negative | Fungi | |||
| Escherichia coli | 1 | Candida albicans | 2 | ||
| Candida sake | 1 | ||||
| Ventilator-associated pneumonia | |||||
|---|---|---|---|---|---|
| Gram-positive | Gram-negative | Fungi | |||
| Serratia marcescens | 1 | ||||
| Mediastinitis | |||||
|---|---|---|---|---|---|
| Gram-positive | Gram-negative | Fungi | |||
| Coagulate-negative Staphylococcus spp. | 1 | Trichosporon inkin | 1 | ||
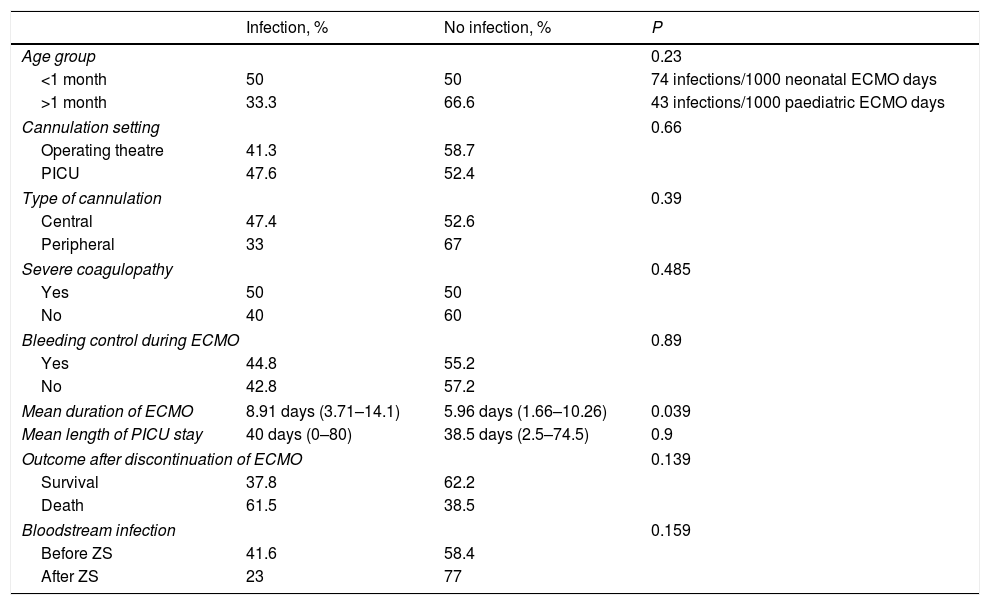
We analysed several variables to determine whether certain characteristics are associated with an increased risk of infection. We compared the proportion of infection by age group (newborns vs age >1 month), the setting where cannulation was performed (operating theatre vs PICU), the type of cannulation (central vs peripheral), whether the patient suffered severe coagulopathy (requiring multiple transfusions) or required some type of intervention during ECMO (surgical bleeding control, repeat surgery, catheterization). The proportion of infection was slightly greater in patients aged less than 1 month, when cannulation was performed in the PICU, with central cannulation, in patients with severe coagulopathy and in patients that required invasive intervention during ECMO, but none of these differences were statistically significant. We also compared the duration of ECMO, mean length of PICU stay and survival after discontinuation of ECMO in patients that developed infection during ECMO vs patients that did not. Patients that developed infection received ECMO support for a longer period (statistically significant difference, P=.039), and their survival was lower, although this difference was not statistically significant. The mean length of stay in the PICU was similar in both groups. In our unit, we introduced a Zero Septicaemia protocol in December 2012, and 24 of the cases of ECMO support occurred before its introduction, while 26 occurred after it. The proportion of bloodstream infection decreased after the implementation of this set of preventive measures, although this decrease was not statistically significant. Table 3 provides detailed information on these proportions and the corresponding P-values.
Description of variables based on the development of infection.
| Infection, % | No infection, % | P | |
|---|---|---|---|
| Age group | 0.23 | ||
| <1 month | 50 | 50 | 74 infections/1000 neonatal ECMO days |
| >1 month | 33.3 | 66.6 | 43 infections/1000 paediatric ECMO days |
| Cannulation setting | 0.66 | ||
| Operating theatre | 41.3 | 58.7 | |
| PICU | 47.6 | 52.4 | |
| Type of cannulation | 0.39 | ||
| Central | 47.4 | 52.6 | |
| Peripheral | 33 | 67 | |
| Severe coagulopathy | 0.485 | ||
| Yes | 50 | 50 | |
| No | 40 | 60 | |
| Bleeding control during ECMO | 0.89 | ||
| Yes | 44.8 | 55.2 | |
| No | 42.8 | 57.2 | |
| Mean duration of ECMO | 8.91 days (3.71–14.1) | 5.96 days (1.66–10.26) | 0.039 |
| Mean length of PICU stay | 40 days (0–80) | 38.5 days (2.5–74.5) | 0.9 |
| Outcome after discontinuation of ECMO | 0.139 | ||
| Survival | 37.8 | 62.2 | |
| Death | 61.5 | 38.5 | |
| Bloodstream infection | 0.159 | ||
| Before ZS | 41.6 | 58.4 | |
| After ZS | 23 | 77 | |
ZS, zero septicaemia protocol.
Nosocomial infections are a frequent complication in patients managed with ECMO. The reported incidence of infection varies widely between studies, from 8.5% in a study in adults requiring ECMO for any indication conducted in Taiwan by Hsu et al.4 to 64% in a study conducted in Paris by Schmidt et al. in adult patients with cardiogenic shock that required veno-arterial ECMO.2 The last report of the ELSO Registry described an overall incidence of infection of 11.7%, with a higher incidence in paediatric patients and adults compared to newborns.3 In our study, the incidence of infection was high (40%, with a 50% incidence in newborns and a 33.3% in paediatric patients), evincing a pressing need to introduce measures for improvement.
Based on the current literature, bloodstream infection and ventilator-associated pneumonia are the most frequent types of infection that develop in patients under ECMO.7,8 In our study, the most frequent type of infection was bloodstream infection (70% of infections).
The most frequent causative agents were coagulase-negative staphylococci, followed by various Enterobacteriaceae and Candida spp., which was consistent with the existing literature, as can be seen in the 2011 report of the ELSO Registry, in which coagulase-negative staphylococci were the most frequent cause of infection (16% of infections) followed by Candida species (12.7% of infections),3 or in the study conducted in Melbourne in 2013 by Aubron et al., where Enterobacteriaceae were the most frequent cause of infection overall (16 out of 46 infections) and Candida organisms the most frequent cause of bloodstream infection (9/24).7
In our study, we found that the duration of ECMO was significantly longer in patients that developed infection compared to patients that did not (8.91 days [3.71–14.1] vs 5.96 days [1.66–10.26] of patients without infection; P=.039), which was consistent with the findings of other studies, such as the one published by Schmidt et al., who concluded that the development of infectious complications was associated with increased duration of ECMO (16±17 days in patients with at least 1 infection vs 8±5 days in patients with no infections; P<.0001),2 or the analysis by Adam et al. of the data of the ELSO Registry, which led them to conclude that increased ECMO is a risk factor for infection in every age group (in newborns, the mean duration of ECMO was 10.61±7.27 days in the group with infection vs 6.66±4.96 days in the group without infection [P<.001]; in the paediatric age group, the mean duration was 13.38±10.55 days in the group with infection vs 7.16±6.75 days in the group without infection [P<.001]).6 This could reflect that patients that develop some type of infection end up requiring ECMO for a longer time, or that greater duration of ECMO carries an increased risk of infectious complications.
Our study did not find any significant associations of infection with other variables, such as central vs peripheral cannulation. Some studies in the literature have analysed the differences between central and peripheral cannulation in ECMO in terms of the incidence of bleeding, repeat surgery or patient outcomes,10 but we found only 1 study that analysed this variable specifically in relation to infection, the study by Schmidt et al., which also found no significant differences although, as occurred in our study, found a higher proportion of infection in patients with central cannulation (infection in 65% of patients with central cannulation vs 45% of patients with peripheral cannulation; P=.71).2
The proportion of infection was greater in patients that died after removal of ECMO (61.5% vs 37.8% in survivors), although the difference was not statistically significant. Data on infection and mortality differ between research groups. The Melbourne group, which conducted a study in 139 patients, concluded that while the incidence of infection in patients under ECMO was associated with an increase in length of PICU stay, it was not associated with an increase in mortality,7 a conclusion that was also reached by Coffin et al. in Philadelphia in a study of 80 newborns that required ECMO.11 However, the 2011 report on the ELSO Registry, which analysed data for 20.741 cases of ECMO support, described a mortality of 57.6% in patients that developed infection compared to 41.5% in patients that did not develop infection (P<.001).3
Early detection of infectious complications is important, yet it is challenging, as the signs and symptoms of infection are difficult to interpret in patients under ECMO. These patients may have manifestations such as hypotension, coagulopathy, thrombocytopaenia, elevation of acute phase reactants and fluctuations of body temperature in the absence of infection.12 Prophylactic antibiotherapy and performance of serial cultures may be useful in these patients. The retrospective study of Kaczala et al., in which daily cultures were performed routinely in ECMO patients and antibiotic prophylaxis was not used in any, detected infections in 22.6% of patients. They concluded that monitoring by means of daily blood cultures is preferable than routine antibiotic prophylaxis because it helps prevent the emergence of drug-resistant organisms.13 On the other hand, another study that was conducted in 4 ECMO centres by Elerian et al. (University of Texas) and included patients that received antibiotic prophylaxis and were monitored with daily culture concluded that routine culture was not indicated and unnecessarily increased health care costs in patients in whom infection was not suspected.14 In our unit, we reviewed existing guidelines for antibiotic prophylaxis and did not find specific recommendations for its use in the context of ECMO (as to the specific antibiotic agents that should be use or the duration of therapy),15,16 and decided to apply the same protocol used in patients after cardiac surgery, using the same recommended drugs for a total duration of 24h, and closely watching patients for any signs of infection.
There are limitations to this study. The main one is its retrospective design, which made it difficult to differentiate the cases in which antibiotherapy was administered in response to a positive culture or rather maintained as a precaution due to the critical condition of the patient. Furthermore, many samples for culture were collected when the patient was already receiving antibiotics, which may have been the reason for some negative results, and in some cases antibiotherapy may have been maintained due to a strong suspicion of infection despite negative results of culture. Thus, we are unable to determine whether our data overestimates or underestimates the actual incidence of infection. In addition, we excluded patients with infections developed prior to ECMO initiation, and thus were unable to assess whether ongoing antibiotic treatment at the time of ECMO initiation was associated with a decreased incidence of bacterial infection or an increased risk of fungal infection during ECMO. Another limitation is the small sample size of our study, which may be the reason why many of our results were not statistically significant, limiting our ability to draw conclusions as to the changes that could be implemented in clinical practice to reduce the incidence of nosocomial infection in these patients.
The incidence of infection during ECMO is very high, and in our study, the only variable that was significantly associated with a higher incidence of infection was longer duration of ECMO, which highlights the importance of improving the condition of the patient as fast as possible to minimise the duration of ECMO. In these patients, strict adherence to the Zero Septicaemia, Zero Pneumonia and Zero Resistance protocols is essential to prevent infection, with especial emphasis on ensuring that any interventions performed on the patient by other specialists (cardiologists, heart surgeons, radiologists, etc.) conform to adequate aseptic and, when necessary, sterile procedure (catheter handling, investigation of mediastinal bleeding, etc.). Measures taken to raise awareness in the entire staff involved in the care of these patients should include regular trainings on handwashing technique, reminders of Zero protocols and meetings to inform on the incidence of infection in the unit.
Considering the importance of nosocomial infection in patients supported with ECMO and the difficulty of its diagnosis, it is important to collect samples for culture and initiate antibiotherapy while awaiting its results in patients that exhibit worsening of symptoms or of laboratory parameters. Given the current lack of standardised protocols or approaches to the use of antibiotic prophylaxis or routine performance of culture in these patients, further studies are needed, preferably with a multicentre design, to establish the optimal approach.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Calderón Checa RM, Rojo Conejo P, González-Posada Flores AF, Llorente de la Fuente AM, Palacios Cuesta A, Aguilar JM, et al. Infecciones durante oxigenación de membrana extracorpórea. An Pediatr (Barc). 2018;89:86–91.
Previous presentation: this study was presented at the XXX Congress of the Sociedad Española de Cuidados Intensivos Pediátricos, May 7–9, 2015.