Neonatal seizures (NSs) occur in 1.5 per 1000 live births, with a higher incidence in preterm newborns. They usually result from asphyxia, haemorrhage, brain malformations, metabolic disturbances, inborn errors of metabolism or infection.1 The challenges posed by its diagnosis and the decision of which children to treat are compounded by the growing evidence on the deleterious effects of the drugs approved for this purpose: phenobarbital and phenytoin. Both achieve seizure reduction in at least 50% of cases, and have been associated with neuronal apoptosis in animal models. For this reason, other drugs that have fewer apparent side effects are increasingly being used off-label. Levetiracetam (LEV) was approved by the Food and Drug Administration in 20122 for the treatment of partial-onset seizures in children aged 1 month and older.
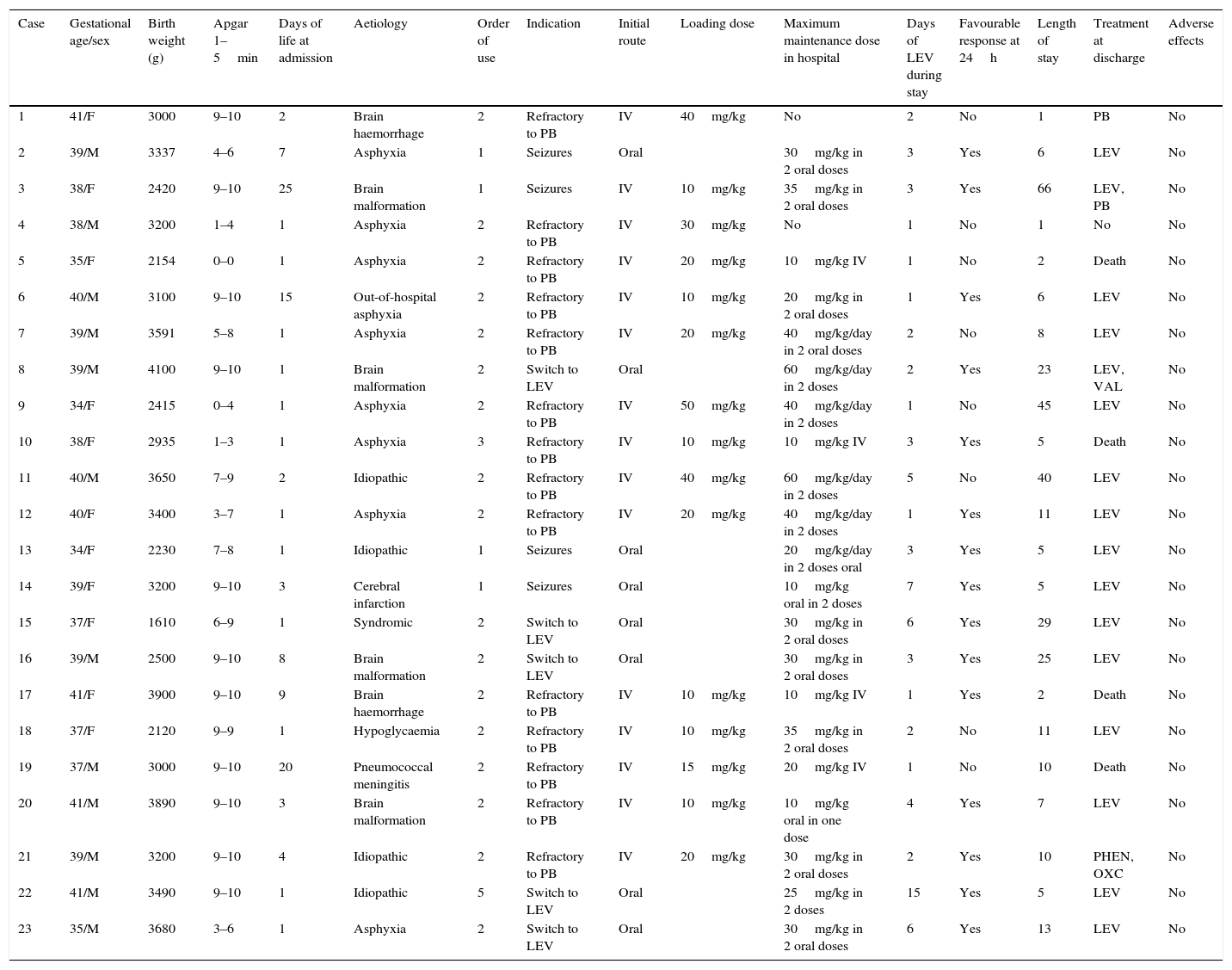
We reviewed the use of LEV in newborns admitted to our unit between January 2011 and May 2016. Table 1 summarises the characteristics of these patients. Twenty-three newborns were treated. Their weights ranged between 1610 and 4100g, and 17.4% had been born preterm. Table 2 presents data on the types of seizures, diagnostic tests and outcomes.
Patient characteristics.
| Case | Gestational age/sex | Birth weight (g) | Apgar 1–5min | Days of life at admission | Aetiology | Order of use | Indication | Initial route | Loading dose | Maximum maintenance dose in hospital | Days of LEV during stay | Favourable response at 24h | Length of stay | Treatment at discharge | Adverse effects |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 41/F | 3000 | 9–10 | 2 | Brain haemorrhage | 2 | Refractory to PB | IV | 40mg/kg | No | 2 | No | 1 | PB | No |
| 2 | 39/M | 3337 | 4–6 | 7 | Asphyxia | 1 | Seizures | Oral | 30mg/kg in 2 oral doses | 3 | Yes | 6 | LEV | No | |
| 3 | 38/F | 2420 | 9–10 | 25 | Brain malformation | 1 | Seizures | IV | 10mg/kg | 35mg/kg in 2 oral doses | 3 | Yes | 66 | LEV, PB | No |
| 4 | 38/M | 3200 | 1–4 | 1 | Asphyxia | 2 | Refractory to PB | IV | 30mg/kg | No | 1 | No | 1 | No | No |
| 5 | 35/F | 2154 | 0–0 | 1 | Asphyxia | 2 | Refractory to PB | IV | 20mg/kg | 10mg/kg IV | 1 | No | 2 | Death | No |
| 6 | 40/M | 3100 | 9–10 | 15 | Out-of-hospital asphyxia | 2 | Refractory to PB | IV | 10mg/kg | 20mg/kg in 2 oral doses | 1 | Yes | 6 | LEV | No |
| 7 | 39/M | 3591 | 5–8 | 1 | Asphyxia | 2 | Refractory to PB | IV | 20mg/kg | 40mg/kg/day in 2 oral doses | 2 | No | 8 | LEV | No |
| 8 | 39/M | 4100 | 9–10 | 1 | Brain malformation | 2 | Switch to LEV | Oral | 60mg/kg/day in 2 doses | 2 | Yes | 23 | LEV, VAL | No | |
| 9 | 34/F | 2415 | 0–4 | 1 | Asphyxia | 2 | Refractory to PB | IV | 50mg/kg | 40mg/kg/day in 2 doses | 1 | No | 45 | LEV | No |
| 10 | 38/F | 2935 | 1–3 | 1 | Asphyxia | 3 | Refractory to PB | IV | 10mg/kg | 10mg/kg IV | 3 | Yes | 5 | Death | No |
| 11 | 40/M | 3650 | 7–9 | 2 | Idiopathic | 2 | Refractory to PB | IV | 40mg/kg | 60mg/kg/day in 2 doses | 5 | No | 40 | LEV | No |
| 12 | 40/F | 3400 | 3–7 | 1 | Asphyxia | 2 | Refractory to PB | IV | 20mg/kg | 40mg/kg/day in 2 doses | 1 | Yes | 11 | LEV | No |
| 13 | 34/F | 2230 | 7–8 | 1 | Idiopathic | 1 | Seizures | Oral | 20mg/kg/day in 2 doses oral | 3 | Yes | 5 | LEV | No | |
| 14 | 39/F | 3200 | 9–10 | 3 | Cerebral infarction | 1 | Seizures | Oral | 10mg/kg oral in 2 doses | 7 | Yes | 5 | LEV | No | |
| 15 | 37/F | 1610 | 6–9 | 1 | Syndromic | 2 | Switch to LEV | Oral | 30mg/kg in 2 oral doses | 6 | Yes | 29 | LEV | No | |
| 16 | 39/M | 2500 | 9–10 | 8 | Brain malformation | 2 | Switch to LEV | Oral | 30mg/kg in 2 oral doses | 3 | Yes | 25 | LEV | No | |
| 17 | 41/F | 3900 | 9–10 | 9 | Brain haemorrhage | 2 | Refractory to PB | IV | 10mg/kg | 10mg/kg IV | 1 | Yes | 2 | Death | No |
| 18 | 37/F | 2120 | 9–9 | 1 | Hypoglycaemia | 2 | Refractory to PB | IV | 10mg/kg | 35mg/kg in 2 oral doses | 2 | No | 11 | LEV | No |
| 19 | 37/M | 3000 | 9–10 | 20 | Pneumococcal meningitis | 2 | Refractory to PB | IV | 15mg/kg | 20mg/kg IV | 1 | No | 10 | Death | No |
| 20 | 41/M | 3890 | 9–10 | 3 | Brain malformation | 2 | Refractory to PB | IV | 10mg/kg | 10mg/kg oral in one dose | 4 | Yes | 7 | LEV | No |
| 21 | 39/M | 3200 | 9–10 | 4 | Idiopathic | 2 | Refractory to PB | IV | 20mg/kg | 30mg/kg in 2 oral doses | 2 | Yes | 10 | PHEN, OXC | No |
| 22 | 41/M | 3490 | 9–10 | 1 | Idiopathic | 5 | Switch to LEV | Oral | 25mg/kg in 2 doses | 15 | Yes | 5 | LEV | No | |
| 23 | 35/M | 3680 | 3–6 | 1 | Asphyxia | 2 | Switch to LEV | Oral | 30mg/kg in 2 oral doses | 6 | Yes | 13 | LEV | No |
F, female; LEV, levetiracetam; M, male; PB, phenobarbital; PHEN, phenytoin; OXC, oxcarbazepine; VAL, valproic acid.
Seizure characteristics and diagnostic tests.
| N (%) | |
|---|---|
| Type of seizures | |
| Clonic | 21 (91.2) |
| Subtle only | 1 (4.4) |
| Tonic | 1 (4.4) |
| Causes | |
| Asphyxia | 9 (39.1) |
| Brain malformation | 5 (21.7) |
| Haemorrhage | 3 (13) |
| Idiopathic | 3 (13) |
| Infection | 1 (4.4) |
| Metabolic | 1 (4.4) |
| Hypoglycaemia | 1 (4.4) |
| Diffusion-weighted MRI | 21 (91.2) |
| Abnormal MRI | 15 (71.4) |
| Head ultrasound only | 2 (8.8) |
| Abnormal | 2 (100) |
| aEEG* | 21 (91.2) |
| Seizures | 17 (81) |
| Abnormal background pattern only | 4 (19) |
| EEG | 22 (95.6) |
| Abnormal | 14 (63.6) |
EEG, electroencephalogram; MRI, magnetic resonance imaging. aEEG*: amplitude-integrated electroencephalogram.
Levetiracetam was used as the first-line agent in 17.4% of the patients, and was the second-line agent in 73.9%, in most cases after phenobarbital failure. In 15 patients (65.2%), treatment was initiated with an intravenous loading bolus at a dose of 10–50mg/kg. A maintenance dose was used in 91.3% that ranged between 10 and 40mg/kg/2 doses, with increases of 10mg/kg every three to five days. Out of the 19 patients in which it was used as the second-line agent, the dose of the first-line agent could be reduced in 63.1% in the first week after initiation of LEV. In eight patients, there was no improvement in either clinical or electrical seizures in the first 24h, but 15 patients (65.2%) did improve (with improvement understood as the cessation of seizures or a reduction in frequency of at least 50%). We did not find evidence of adverse effects in any of the patients. There was treatment limitation in four patients, which was unrelated to the use of LEV. Of the 19 remaining patients, 88.9% were discharged with a LEV regimen (77.8% as monotherapy).
The use of LEV in NSs is becoming increasingly frequent despite not having been approved, as it is the case of most anticonvulsant agents used in infants. It is more recommended by paediatric neurologists than by neonatologists.1,2 A retrospective chart analysis3 identified 72 newborns treated with LEV, most of them born preterm, and found no adverse effects leading to treatment discontinuation. The definition of seizure was based on clinical features only, as aEEG or EEG were not used, so the efficacy results of this study are debatable. Previous studies have included between six and thirty-eight newborns,2 and some were exclusively on preterm neonates (n=12).4 One prospective study5 in 38 newborns in which LEV was used as the first-line agent found clinical improvement, and the EEG taken at one week post birth in 30 of these newborns showed no evidence of side effects, although the protocol tolerated the administration of two doses of phenobarbital during LEV titration, so that phenobarbital or the natural course of the seizures may have influenced the results. There is a published case of anaphylaxis following infusion of LEV in a neonate,6 and it is possible that children with complex clinical situations and previous management are difficult to identify. A recent study found an association between LEV and neuronal cell death in newborns with asphyxia not treated with hypothermia.6 Given the scarcity of the evidence on newborns, the safety of this drug for the first-line treatment of NSs remains to be determined, and most current guidelines that include LEV do so as a second-line agent.
There are limitations to this review. In addition to its retrospective design, the number of cases is small, although we have not found any other Spanish publication on the subject; our unit was not equipped with video-EEG to diagnose seizures, although we used aEEG and EEG to guide the treatment. A larger sample size would help determine whether there is an association between gestational age or the aetiology of the seizures and the response to LEV.
In conclusion, LEV is increasingly being used for the treatment of NSs, usually as the second-line agent. It seems to be at least as effective as classical anticonvulsants. Since it was associated with few side effects and seems to be less harmful to the brain, LEV is a drug to consider, even in preterm newborns. Prospective studies are needed to learn the effects of this drug in the short and long term.
This review had the approval of the hospital's ethics committee.
Please cite this article as: Lloreda-García JM, Fernández-Fructuoso JR, Gómez-Santos E, García-González A, Leante-Castellanos JL. Uso de levetiracetam en crisis convulsivas neonatales. An Pediatr (Barc). 2017;86:286–288.