Antiphospholipid syndrome (APS) is an autoimmune disease characterised by vascular thrombosis—arterial or venous—or recurrent foetal losses associated with the presence of circulating antiphospholipid antibodies detected on two occasions at least 12 weeks apart. It may be primary, lacking an association to other disorders, or secondary to underlying diseases, most frequently systemic lupus erythematosus. Antiphospholipid antibodies are antibodies that target plasma proteins with an affinity for anionic phospholipids, such as lupus anticoagulant and anticardiolipin and anti-β2-glycoprotein antibodies, which we highlight due to their clinical relevance.1
In 1999, the Sapporo criteria were established for the classification of APS, although these are not diagnostic criteria. An update was made in 2006, and while it has been proposed that they be adapted for their use in the paediatric population by eliminating the obstetric criterion,2 this proposed application has yet to be validated (Table 1).
Adaptation for the paediatric population of the current classification criteria for antiphospholipid syndrome in adults.
| Clinical criteria |
| Vascular thrombosis: –≥1 clinical episodes of arterial, venous, or small vessel thrombosis, in any tissue or organ confirmed objectively by validated criteria |
| Laboratory criteria |
| –Anticardiolipin antibody of IgG or IgM isotype in serum or plasma present in medium to high titre (more than 40 units of IgG or IgM, or > 99th percentile) in 2 or more occasions at least 12 weeks apart –Lupus anticoagulant present in plasma on two or more occasions at least 12 weeks aparta –Anti-β2-glycoprotein IgG and/or IgM isotype in serum or plasma in medium or high titre (>99th percentile) on two or more occasions at least 12 weeks apart |
Antiphospholipid syndrome: at least the clinical criterion and one laboratory criterion are met. Classification of APS should be avoided if less than 12 weeks or more than 5 years separate the positive laboratory test and the clinical manifestation.
Detected according to the guidelines of the International Society on Thrombosis and Haemostasis:
–Prolongation of phospholipid-dependent clotting time evinced by a screening test (aTTP, kaolin clotting time, dilute Russell's viper venom time, prothrombin time, Textarin clotting time).
–The clotting time prolongation is not corrected by mixing the sample with normal plasma poor in platelets.
–The clotting time prolongation improves when the same clotting time test is performed with the addition of phospholipids.
–Ruling out of other aetiologies of coagulopathy (such as factor VIII inhibitor or heparin).
Source: Aguiar et al.2
We present the cases of two patients with thrombocytopaenia refractory to conventional treatment and very high titres of antiphospholipid antibodies that showed a rapid and sustained response to the administration of rituximab (RTX).
Patient 1Female adolescent aged 13 years with a history of extremely corticosteroid-dependent thrombocytopaenia refractory to intravenous immune globulin (IVIG), with associated cyclic neutropaenia (minimum, 600 neutrophils/μL) and lymphocytopaenia (minimum, 500/μL). The myelogram, autoantibody tests and serum complement levels were normal. Ten months after onset, in addition to thrombocytopaenia, the patient exhibited a significantly prolonged aTTP (78s; normal range, 26–38s) with positive tests for lupus anticoagulant, anticardiolipin IgG (>160IU/mL) and anti-β2-GPI IgG (>160IU/mL), confirmed 12 weeks later. The associated clinical manifestations were joint pain and headache. The results of the physical examination, neurologic assessment and imaging tests were normal.
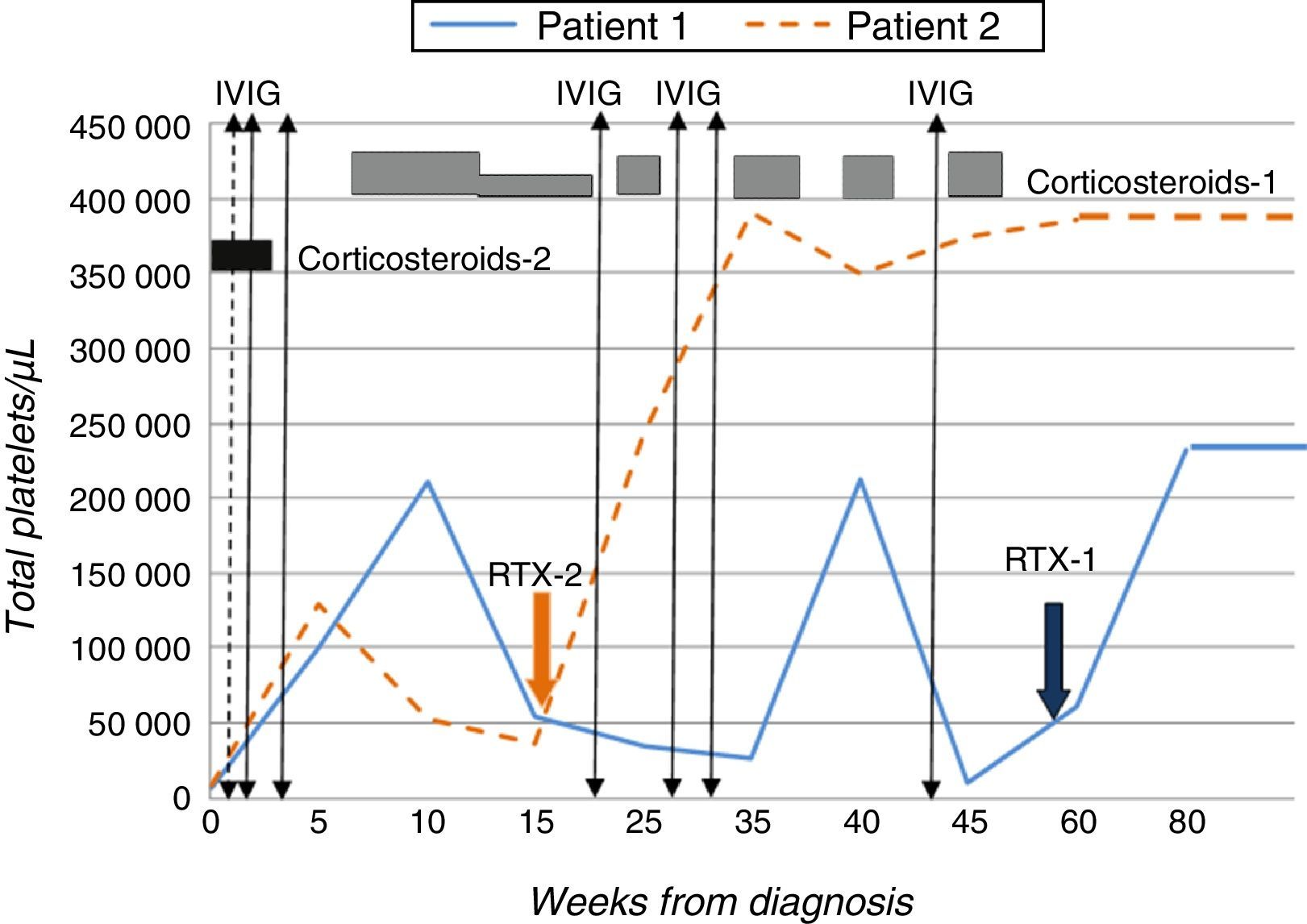
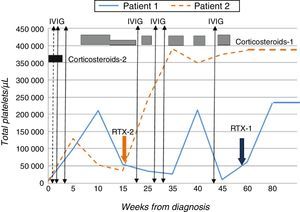
Given the lack of response to conventional treatment, and with a baseline count of 10000platelets/μL, treatment with RTX3 was initiated at a dose of 375mg/m2/week for a period of four weeks, which the patient tolerated well. The level of platelets normalised from the third week of treatment (>140000/μL) (Fig. 1).
Patient 2Male adolescent aged 14 years presenting with purpura and ecchymosis in the context of thrombocytopaenia (8000platelets/μL), prolonged aTTP (>100s) and presence of lupus anticoagulant and anticardiolipin IgG (>160IU/mL) and anti-β2-GPI IgG (>160IU/mL) antibodies that was confirmed 12 weeks later. He received two doses of prednisone, with no response, followed by IVIG, which resulted in a transient increase in platelet levels. In light of the patient's progress and past history, and with platelet counts as low as 30000/μL, treatment with RTX at the dose noted above was initiated, to which the patient responded favourably (Fig. 1).
Neither patient developed thrombotic events, livedo reticularis, Raynaud syndrome or symptoms compatible with lupus erythematosus or other systemic autoimmune disease, nor had haemorrhagic complications during their followup. The cardiologic and ophthalmologic assessments and abdominal ultrasound performed prior to initiation of RTX were also normal.
Given the risk of thrombotic effects, both patients started anticoagulant therapy with acetylsalicylic acid (100mg/day) once it had been confirmed that their platelet levels had stabilised.2,4 At 20 and 10 months of followup, respectively, and after completion of a single course of RTX, both patients continued to have normal platelet counts, with no associated adverse effects and decreasing titres of antiphospholipid antibodies.
Although thrombosis, especially recurrent venous thrombosis, is the most characteristic manifestation of APS, there are other well-defined “non-classical” clinical manifestations of the disease4 that are not included in the classification criteria. They include neurologic changes (migraine, chorea), cutaneous manifestations (livedo reticularis, Raynaud syndrome), heart valve disease, renal microangiopathy and haematologic disorders (immune thrombocytopenia, haemolytic anaemia). These “non-criteria” findings are more frequent in the paediatric population compared to adults,1 unlike thrombotic manifestations.2
The first line of treatment for immune thrombocytopaenia (corticosteroids alone or combined with IVIG) is effective in 60% to 80% of patients. The alternative treatments proposed for refractory cases include anti-D immunoglobulin (Rh+ patients) or splenectomy, but they have limitations, and therefore new therapeutic options need to be investigated.3,5
RTX is a chimeric monoclonal antibody that binds the CD20 antigen. It produces a quick and lasting elimination of peripheral B lymphocytes, and can be used to treat antibody-mediated immune disorders including autoimmune thrombocytopaenia.6 It remains unknown whether lymphocyte B reconstitution would require repetition of the treatment.
The evidence available to date has shown an adequate efficacy and safety, although the use of RTX requires close monitoring due to its potential adverse effects,5,6 which, while rare, are severe—for instance, progressive multifocal leukoencephalopathy.
To conclude, as the use of RTX for the treatment of refractory thrombocytopaenia associated with antiphospholipid antibodies in paediatric patients grows, it is becoming apparent that this is an effective alternative.5,6 Its early use could prevent the adverse effects that result from the prolonged use of corticosteroids, among others.
Please cite this article as: Vázquez Gómez F, Prieto Arce M, González-Granado LI, Enríquez Merayo E, de Inocencio Arocena J. Uso de rituximab para trombocitopenia refractaria en pacientes con anticuerpos antifosfolípidos. An Pediatr (Barc). 2017;87:51–53.